Abstract
Complex anal fistula is a common and refractory anorectal disease in clinical practice.
Surgery is currently the main treatment method, and the sphincter-preserving surgical
approach is the current surgical trend for complex anal fistula. This article reviews
the surgical methods for complex anal fistulas that preserve the sphincter, including
transanal opening of intersphincteric space, ligation of the intersphincteric fistula
tract, loose-seton technique, endorectal advancement flap, mesenchymal adipose-stem cell
transplantation, fistula-tract laser closure, video assisted anal fistula treatment,
fibrin glue and anal fistula plug. This article reviews the articles on minimally
invasive treatment for anal fistula in PubMed, Cochrane Library CENTRAL and Embase, and
introduces the current research progress. The aim is to strive to maintain a balance
between curing complex anal fistulas and protecting the function of the sphincter.
Keywords: Anal Fistula; Sphincter Preservation; Mesenchymal Stem Cells; Laser Therapy; Endoscopic Surgery
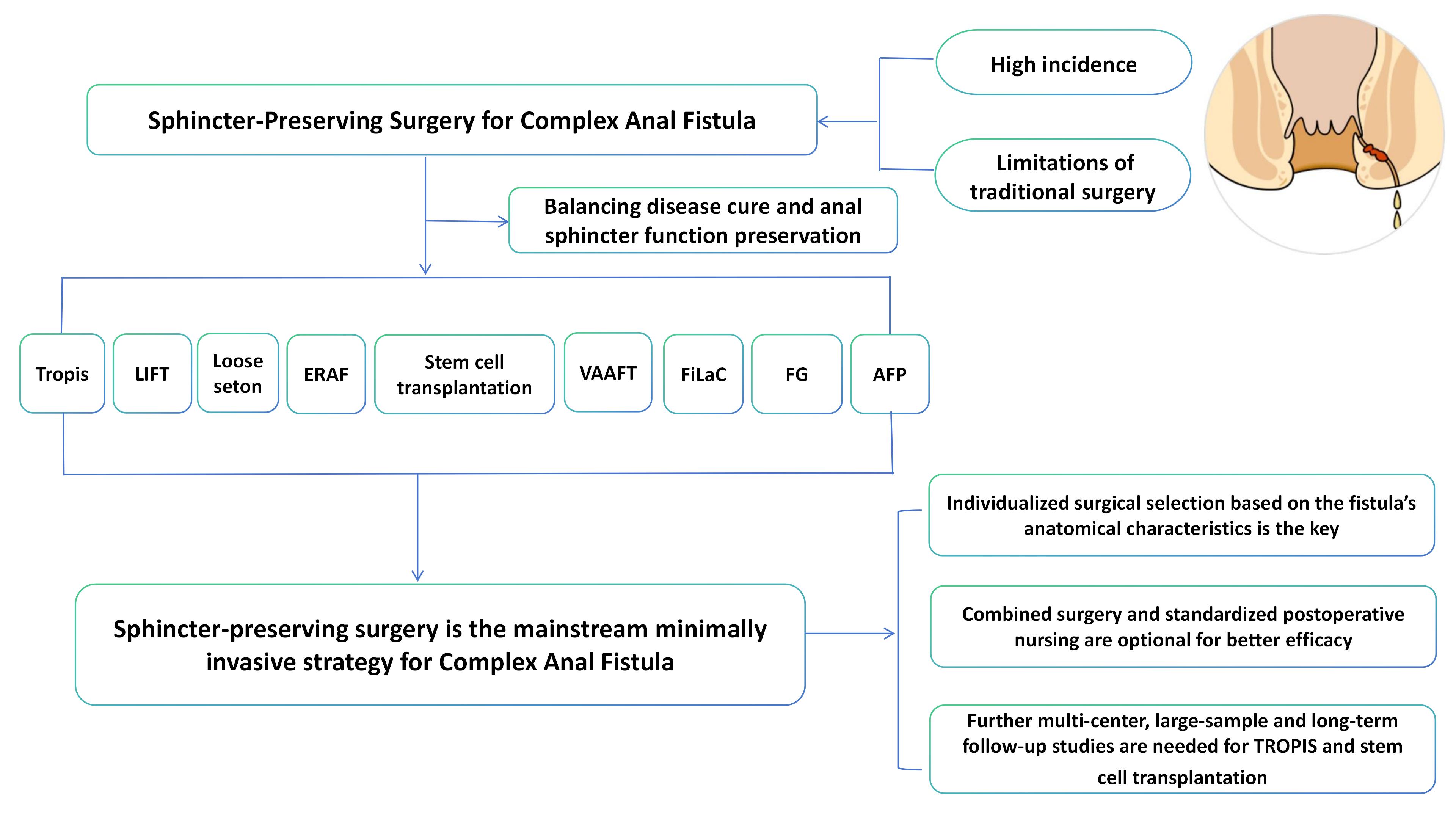
Introduction
Complex anal fistula refers to an anal fistula disease with two or more external and internal openings and two or more branch pipes or fistulas. It is one of the common anorectal surgical diseases in clinical practice. With the increase of work pressure and the change of living habits, the incidence of complex anal fistula has been increasing year by year. Data show that the incidence of complex anal fistula accounts for 1.67% to 3.6% of the incidence of anorectal fistula in China [1] , and 8% to 25% abroad. Its main manifestations are repeated ulceration and discharge of pus from the anus, pain, itching, and prolonged non-healing, which affects the quality of life of patients. At present, surgery is still the preferred method for the clinical treatment of complex anal fistula. Conventional anal fistula resection has problems such as many postoperative complications and a high recurrence rate [2] . Therefore, how to cure complex anal fistulas while protecting the anal function of patients has important clinical significance. With the rise of the concept of minimally invasive treatment, the sphincter-preserving surgical approach has become the mainstream surgical method for treating complex anal fistulas. The surgical methods for preserving the sphincter in complex anal fistulas are summarized as follows:
Tropis: Transanal opening of intersphincteric space
Transsphincter fistula incision was first proposed by Indian scholar Grag in 2017. The key points of this surgery lie in incising the internal orifice and the internal sphincter, opening the sphincter space, and maintaining smooth incision drainage. After long-term follow-up of 61 patients with high and complex anal fistulas, Grag successfully cured 47 patients in the end. The overall cure rate after the operation was 90.4% [3] . Pankaj Garg found that by applying the Tropis procedure to high and complex anal fistulas, the Tropis procedure could maintain the shape of the anal canal and the function of the sphincter to the greatest extent [4] . The postoperative pain was mild, the wound recovery was fast, and the postoperative scar was small, achieving a balance between curing anal fistulas and protecting anal function. It is worthy of clinical application and promotion. Shrivats Mishra treated 35 patients with anal fistulas with Tropis surgery and conducted a 3-month follow-up. Among them, 29 patients achieved complete healing, with a cure rate as high as 82.89% [5] . At present, the specific efficacy of Tropis surgery in the treatment of complex anal fistulas still lacks multi-center and large-sample data support for verification. However, due to its advantage of effectively preserving the sphincter, it is still favored by surgeons.
LIFT: Ligation of the intersphincteric fistula tract
Ligation of the intersphincteric fistula tract was first proposed by Thai doctor Rojanasakul in 2007 [6] . The main operational points are to make an incision between the sphincters, ligate the fistula near the internal sphincter, separate the external opening to the external sphincter fistula, close the defect of the external sphincter, and suture the intersphincter incision. Rojanasakul was the first to apply this surgical method to 18 patients with anal fistula. After a 3-month follow-up, the cure rate was as high as 94.4%. Hariprasad compared the clinical efficacy of 40 patients with anal fistula treated by LIFT and fistula resection and found that LIFT surgery for complex anal fistula has the advantages of less surgical trauma, faster recovery, less pain and lower recurrence rate [7] . Zhang conducted a clinical efficacy analysis on 82 patients with complex anal fistula treated with LIFT [8] . After 4 to 120 months follow-up, 6 cases recurred and formed intersphincter fistulas after the operation, and 9 cases developed persistent or recurrent fistulas after the operation. The total clinical cure rate was 80.5%. For colorectal surgeons, the key to the success of LIFT surgery lies in the correct judgment of the fistula shape. The reasons for its failure or recurrence can be attributed to: (1) incomplete treatment of the internal opening; (2) complex branched sinus tracts; (3) multiple anal surgical treatments have been performed; (4) the infected glands and some sources of infection have not been cleared. However, compared with traditional surgical methods, the LIFT procedure, as a surgical approach through the intersphincter, causes less damage to the sphincter, can protect the anal function to the greatest extent, has a lower probability of postoperative complications, and has a high application value in the treatment of complex anal fistulas.
Furthermore, the combination of biological dressings (BioLIFT) and anal fistula plugs (Lift-plug) with the traditional LIFT surgical method has also been innovatively applied in the surgical treatment of complex anal fistulas with sphincter preservation. Although studies have shown that these two surgical methods have a high cure rate, can effectively protect the anal sphincter, and reduce the recurrence rate, due to the high surgical cost, the therapeutic effect has not been verified by large sample data, so it has not been widely used in clinical practice.
Loose-seton technique
During the operation, the internal orifice and internal sphincter are incised, the primary infection focus is removed, the external sphincter is not resected, and the fistula outside the sphincter is removed for loose-seton [9] . During the process, the external sphincter is not severed. It only serves the functions of drainage, marking, and promoting fibrosis through foreign body stimulation. Therefore, this surgical method has both the characteristics of drainage and the preservation of the sphincter. Kennedy and Zegarra treated 32 patients with relaxed suture drainage [10] . The time for removing the rubber band was 2 to 16 weeks (with an average of 7 weeks), and the average follow-up was 36 months. The cure rate was 78%. Some patients had mild air leakage or overflow, and only 1 case (3%) had occasional loose stool incontinence. No patients had solid stool incontinence.
The loose-seton technique reduces the damage to the sphincter and ensures smooth drainage at the same time. It has a good therapeutic effect on the upper sphincter and the high transsphincter anal fistula. However, there is still some controversy over the cure rate of this surgery at present. When this surgical method is used in clinical practice, it is found that: (1) The timing of removing the double-stranded rubber bands is particularly crucial. Observe that there is no secretion from the wound, the fluid flowing out of the incision is clean after rinsing, and the base of the wound is long and solid. Remove the rubber bands in batches when there is resistance when rotating them; (2) After the operation, the wound surface should be rinsed with normal saline to keep it fresh and clean; (3) Regularly explore the wound surface to prevent false healing; (4) When fibrosis at the incision site in the later stage is difficult to heal, tight suture therapy should be combined to ensure the therapeutic effect.
ERAF: Endorectal advancement flap
The endorectal advancement flap was the first method for treating anal fistula with sphincter preservation, proposed by Elting in 1912 [11] . This method was first used in the treatment of rectovaginal fistula. In 2022, ASCRS "Guidelines for the Treatment of Anal Fistula, Perianal Abscess and Rectovaginal Fistula" recommended a grade of 1B for it, and it has gradually become a relatively mature sphincter-preserving surgical method [12] . The key points of this surgical procedure lie in completely stripping the internal orifice and the surrounding lesion tissues, and creating a "U"-shaped flap (including the mucosa, submucosa and part of the circular muscle, with the bottom wider than the top). After loosening the mucosal flap, it is tensively and intermittently sutured with the surrounding tissues, and the external orifice and the lateral fistula are removed in a tunnel manner. Multiple research reports have shown that the cure rate of rectal mucosal flap displacement in patients with anal fistula ranges from 66% to 87% [13~15] . Carlos Chaveli Diaz performed surgery and follow-up on 115 patients with anal fistula [16] . As a result, 84 patients were cured, and no severe anal function impairment occurred in all patients who adopted this surgical method.
ERAF has a definite therapeutic effect on complex anal fistulas. From a technical perspective, this surgical method has the advantages of not damaging the sphincter, having a relatively small impact on anal function, not affecting the appearance of the anus, causing mild pain, and not affecting reoperation after failure. It has also been widely used for rectovaginal fistulas and rectourethral fistulas. However, the surgical operation is rather complex and requires surgeons with rich clinical experience to carry out. The related influencing factors of its failure and recurrence still require further analysis in large-sample, multi-center clinical studies to better reduce risks and improve surgical efficacy.
Mesenchymal adipose-derived stem cell transplantation
Stem cell transplantation therapy involves transplanting healthy stem cells into a patient's body to repair or replace damaged cells or tissues, thereby achieving the goal of curing diseases [17] . Mesenchymal adipose-derived stem cells are stem cells derived from fat. They are easy to obtain, have a large reserve in the body, and have functions such as tissue regeneration and repair, and inhibition of inflammatory responses. The advantages include no damage to the sphincter, less trauma, and less pain in the treatment of complex anal fistulas [18] . Zhang treated 24 patients with complex anal fistula with autologous adipose-derived stem cell transplantation [19] . They observed indicators such as the cure rate, external opening closure time, anal incontinence score, pain score, and the score of life quality. After regular follow-up, the results showed that after adipose-derived stem cell treatment, the patients had a high quality of life, the anal sphincter was well protected, and no anal incontinence occurred, superior to traditional surgical methods. Panes conducted a randomized, double-blind, parallel, placebo-controlled Phase III clinical trial in 2016, the results showed that the cure rate of the allogeneic adipose-derived stem cell treatment group was significantly better than that of the placebo group, with a lower recurrence rate and a lower incidence of related adverse events [20] .
In addition, several studies have also confirmed that adipose-stem cell transplantation has the advantages of safety, no incision of normal tissues, less damage to anal function, good tolerance, repeatability, quick postoperative recovery, mild pain, and high perioperative quality of life for patients. It is an effective method for treating complex anal fistulas [21,22] . However, the current research sample size is relatively small, and large-sample clinical research data is needed to confirm its efficacy.
Other sphincter-preserving surgical methods
Video-assisted anal fistula treatment (VAAFT) combines endoscopic techniques with the concept of sphincter preservation. Its surgical advantage lies in the ability to explore and handle the fistula and internal orifice under direct video vision [20] , to remove the necrotic tissue of the fistula, and to close the inner opening without damaging the anal sphincter. Siddique believes that VAAFT has the advantages of less intraoperative bleeding, less postoperative pain, less anal sphincter injury, faster wound healing and shorter hospital stay in the treatment of complex anal fistulas, making it a better surgical method for the treatment of complex anal fistulas [23] . KOCHHAR treated 82 patients with anal fistula with VAAFT from 2010 to 2014, with a cure rate of 84% [24] . In this study, anal canal manometry was used to evaluate the sphincter function of patients before and after the operation. The results showed that there was no statistically significant difference in the resting and systolic pressure of the anal canal between patients before and after the operation (P>0.05), indicating that the sphincter function was not damaged, confirming that VAAFT has a relatively optimistic clinical application prospect in the treatment of complex anal fistulas.
Fistula-tract laser closure (FiLaC) is a new sphincter-preserving surgical method. It causes the tissues inside the fistula to vaporize and contract through the energy released by the circular laser at the tip of the catheter, thereby closing the fistula. Foreign studies have shown that the cure rate of this method for treating anal fistula is 71%-82% [25,26] . While curing anal fistula, patients have mild postoperative pain, and no fecal incontinence occurs, and the anal function is maximized, indicating that this is a safe and effective surgical method.
The anal fistula plug and fibrin glue, as new biological scaffold materials, have gradually been applied in the treatment of complex anal fistulas. Anal fistula plug is an absorbable material made from pig small intestines. When placed in the patient's body, it can rapidly generate new blood vessels and promote the reproduction of fibroblasts, thereby achieving the effect of repair. Studies have confirmed that anal fistula plugs have significant effects in closing the internal opening and filling fistulas [27] . They can accelerate the healing time of patients' wounds, cause no major trauma to patients, and have no impact on the appearance and function of the patient's anus. Fibrin glue is a natural extracellular matrix with the advantage of good histocompatibility. It can promote the formation of vascular scars and stimulate the proliferation of fibroblasts and the formation of collagen fibers at the wound site, accelerating wound healing. Moreover, fibrin glue is simple and can be repeatedly implanted. Studies showed that the cure rate of fibrin glue in the treatment of anal fistula was 14% to 63%. Its effect is influenced by multiple factors, but its natural protection of the sphincter and its non-impact on subsequent other treatment methods are still worthy of selection. However, the recurrence rate after anal fistula plug and fibrin glue is relatively high, the long-term effect is not satisfactory, and there is a lack of a large amount of clinical data to support its therapeutic effect. At present, it has not been widely used in clinical treatment.
Table of research results related to sphincter-preserving surgery
A total of 9 articles on sphincter-preserving surgery were included [3,20,28~33] , involving 610 patients, including 5 randomized controlled trials and 4 cohort studies. The included studies summarized the cure rate, and complications of each technique. The results of the included studies are shown in Table 1.
Table 1. The cure rate, and complications of sphincter-preserving surgery.
|
Surgical method |
Study |
Type of study |
Cure rate |
Complications |
Tropis |
Pankaj Garg et al. [3] |
Cohort study |
90.4% |
15.4% |
LIFT |
Tsang et al. [28] |
RCT |
87.5% |
12.5% |
Loose-seton technique |
Kennedy et al. [10] |
RCT |
78% |
3% |
Stem cell Transplantation |
Panés et al. [20] |
RCT |
50% |
17% |
AF |
Mujukian et al. [29] |
Cohort study |
65% |
8% |
VAAFT |
Cheung et al. [30] |
RCT |
81% |
12.9% |
FiLaC |
Wilhelm A et al. [31] |
Cohort study |
64.1% |
2.5% |
FG |
Haim N et al. [32] |
RCT |
63.04% |
4.34% |
AFP |
Yu Tao et al. [33] |
Cohort study |
56% |
35% |
The cure rate, and complications of sphincter-preserving surgery. Tropis, transanal opening of intersphincteric space; LIFT, ligation of intersphincteric fistula tract; AF, advancement flap; VAAFT, video-assisted anal fistula treatment; FiLaC, fistula-tract laser closure; FG, fibrin glue; AFP, anal fistula plug.
Conclusion
At present, with the advancement of medical technology and the improvement of medical humanistic care, the treatment of complex anal fistulas is no longer simply limited to treatment. How to ensure the anal function of patients and guarantee their life quality while achieving cure is the focus of clinicians' attention. The emergence of various sphincter-preserving surgeries has further improved the therapeutic effect of complex anal fistulas, and can minimize the risk of postoperative complications for patients to the greatest extent, which is of great significance for improving the quality of life of patients. Clinically, the appropriate surgical method can be selected based on the fistula condition of the patient. When necessary, other surgical methods can be combined, along with postoperative care, to achieve the goal of curing anal fistula without damaging the sphincter and anal function, and to improve the long-term efficacy of the sphincter-preserving surgical method for anal fistula.
Abbreviations
Advancement flap: AF; Anal fistula plug: AFP; Biological ligation of the intersphincteric fistula tract: BioLIFT; Endorectal advancement flap: ERAF; Fibrin glue: FG; Fistula-tract laser closure: FiLaC; Ligation of the intersphincteric fistula tract: LIFT; Ligation of the intersphincteric fistula tract plug: Lift-plug; Transanal opening of intersphincteric space: Tropis; Video-assisted anal fistula treatment: VAAFT.
Declarations
Author contributions
Ling Wang and Xindan Zhang: writing - original draft. Ji Jin and Fuheng Liu: writing - original modification. Gang Zhao: writing - review & editing. All authors read and approved the final manuscript.
Acknowledgements
Not applicable.
Funding information
This project was supported by Science and Technology Plan of Suqian City (Grant No. S202220).
Ethics approval and consent to participate
Not applicable.
Competing Interests
The authors declare that they have no existing or potential commercial or financial relationships that could create a conflict of interest at the time of conducting this study.
Data Availability
Not applicable.
References
 Figures
Figures References
References Peer
Peer Information
InformationNone.
Peer-review Terminology
Identity transparency: Single anonymized
Reviewer interacts with: Editor
Details
This is an open access article under the terms of the Creative Commons Attribution License(http://creativecommons.org/licenses/by/4.0/), which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Publication History
Received 2025-08-19
Accepted 2025-09-06
Published 2026-03-21


