Abstract
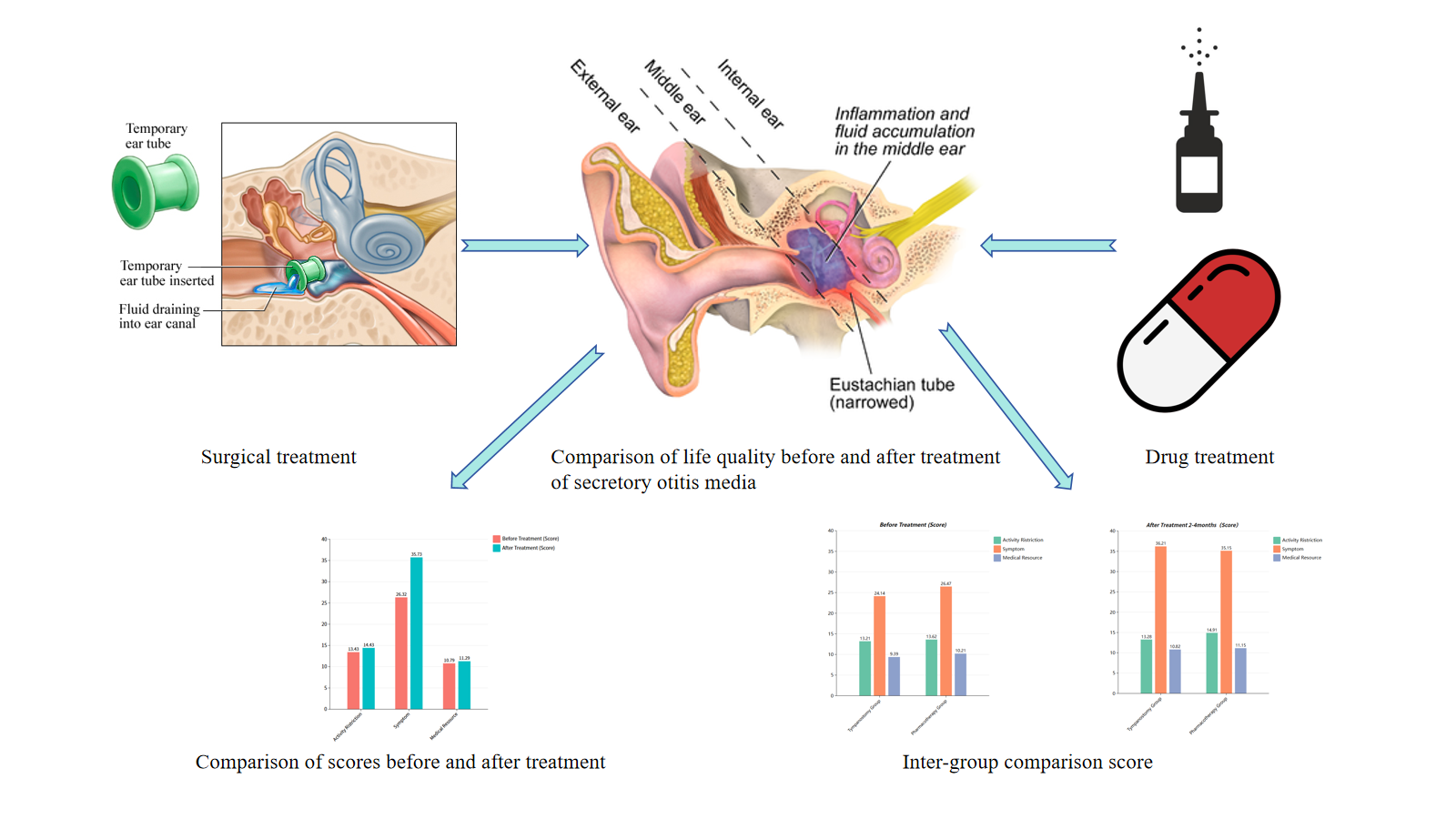
Objective:This study aimed to compare the impact of surgical treatment (tympanostomy tube insertion) and pharmacological therapy on quality of life (QoL) in patients with chronic secretory otitis media (CSOM), focusing on symptom alleviation and functional recovery.
Methods: A prospective, longitudinal, non-randomized study was conducted at Chaohu Hospital, China, involving 80 CSOM patients (aged 6–60 years) divided into surgical (n=38) and pharmacological (n=42) groups. QoL was assessed using a modified Chinese Chronic Ear Survey (CCES) before and 2–4 months post-treatment. The CCES evaluated activity restriction, symptom severity, and healthcare utilization. Statistical analysis via SPSS 28.0 included paired and unpaired t-tests.
Results:Both treatment regimens significantly improved symptom scores (p<0.05), with the surgical group demonstrating marginally superior efficacy (mean symptom score improvement from 24.14 to 36.21 in the surgical group versus 26.47 to 35.15 in the pharmacotherapy group). However, neither intervention showed statistically significant differences in overall quality of life (QoL) improvement (p>0.05). Activity limitation scores showed no significant change following treatment, indicating persistent functional impairment. Demographic factors including gender, educational attainment, and socioeconomic status had no significant influence on treatment outcomes.
Conclusion:While both surgical and pharmacological treatments alleviate CSOM symptoms, neither fully restores daily functioning, underscoring incomplete QoL recovery. These findings highlight the need for long-term evaluations and personalized treatment strategies. Future research should address study limitations, including small sample size and lack of laboratory-based assessments, to refine therapeutic approaches for holistic patient well-being.
Keywords: secretory otitis media, quality of life, tympanostomy tube insertion, medication treatment, comparative study
Introduction
Chronic secretory otitis media (CSOM) is a prevalent middle ear disorder worldwide, characterized by the accumulation of non-purulent fluid in the middle ear cavity, leading to symptoms such as hearing loss, tinnitus, and aural fullness[1] .Although CSOM is particularly common in children, its impact extends beyond this population. Adults, especially those with eustachian tube dysfunction or frequent upper respiratory tract infections, may also develop this condition, significantly compromising their quality of life.Recent epidemiological studies indicate a high incidence of CSOM among children globally, particularly in developing countries. Research in Asia demonstrates notable prevalence rates, with childhood CSOM reaching 7.89% in China, and an overall prevalence of 5.13%[2] .This high incidence not only threatens children's auditory development but may also adversely affect their linguistic and cognitive growth. In contrast, while CSOM is less prevalent in adults, it remains clinically significant. Persistent untreated middle ear effusion in adults can lead to chronic hearing impairment and diminished quality of life.Effective treatment of CSOM is therefore crucial from both public health and individual quality-of-life perspectives. Current therapeutic approaches are broadly categorized into surgical and pharmacological interventions. Surgical treatments, such as tympanocentesis and tympanostomy tube placement, are often considered effective, particularly for recurrent or refractory cases[3] .Pharmacological management, including antibiotics, corticosteroids, and antihistamines, was historically widely used but has faced increasing skepticism due to limited efficacy and concerns about adverse effects[4] .Nevertheless, pharmacotherapy remains an option in specific scenarios, such as when surgical risks are prohibitive or patients decline surgery[5] .Given the substantial impact of CSOM on patients' quality of life and the trade-offs between existing treatments, this study aims to rigorously compare the effectiveness of surgical versus pharmacological interventions in improving quality of life for CSOM patients[6] . Through systematic evaluation and analysis, we seek to provide robust evidence to guide clinical decision-making, ultimately enhancing patient outcomes.
Materials and Methods
Inclusion and Exclusion Criteria
Inclusion Criteria:Age between 6 and 60 years.Confirmed diagnosis of chronic secretory otitis media (CSOM) meeting International Classification of Diseases (ICD) criteria, with complete pure-tone audiometry and tympanometry results.Disease duration exceeding three months, accompanied by symptoms such as tinnitus, hearing loss, or aural fullness.Willingness of patients (or legal guardians) to participate in the study, with signed informed consent.Completion of standardized quality-of-life assessment prior to enrollment, with documented scoring results.
Exclusion Criteria:Presence of other severe otolaryngological conditions (e.g., complications of otitis media, tumors).History of ear surgery within the past six months.Current use (within the past three months) of medications affecting auditory function (e.g., certain antibiotics or diuretics).Diagnosis of severe psychiatric disorders or cognitive impairment impairing comprehension of study objectives or compliance with the protocol.Factors hindering follow-up (e.g., remote residence or noncompliance with study interventions).
General Information
Eighty eligible patients with chronic serous otitis media (CSOM) were prospectively enrolled in this longitudinal, non-randomized study at Chaohu Hospital Affiliated to Anhui Medical University between November 2023 and June 2024.
Study Design and Patient Evaluation
This study employed a questionnaire-based method for data collection from enrolled participants. We recruited eligible individuals diagnosed with chronic secretory otitis media at the Department of Otolaryngology, Chaohu Hospital Affiliated to Anhui Medical University. All participants provided written informed consent after being fully informed about the study. Initial questionnaires were administered before treatment, with follow-up questionnaires conducted 2-4 months post-treatment. Comparative analysis was performed between pre- and post-treatment questionnaire results. Based on treatment modalities, patients were divided into two groups: tympanostomy with tube insertion group and pharmacotherapy group, with outcomes compared between these groups.
Assessment Instruments
This study utilized a modified Chinese version of the Chronic Ear Survey (CCES). The CCES is a subjective questionnaire specifically designed for patients with chronic otitis media, originally developed by the Clinical Outcomes Research Team at Massachusetts Eye and Ear, Harvard Medical School, and later translated into Chinese in 2000 by relevant institutions in Taiwan (including the Department of Otolaryngology at Cathay General Hospital). The original scale consists of 13 questions divided into three subscales: activity restriction, symptoms, and healthcare utilization.(Table 1)[7] .
Table 1. Chronic Ear Survey.
| Questionnaire | |
|
Activity Ristriction (AR) a1.Due to your ear condition, you would not swim or shower without tightly blocking your ears: (1)Absolutely true (2)True (3)Uncertain (4)False (5)Absolutely false
a2.How severely does the need to prevent water from entering your ears currently restrict you? (1)Extremely severe (2)Severe (3)Moderate (4)Mild (5)Very mild (6)No restriction
a3.During the past 4 weeks, has your ear problem affected your social activities with friends, family, or community groups? (1)All of the time (2)Most of the time (3)A good bit of the time (4)Some of the time (5)A little of the time (6)None of the time
Symptom (ST) s1.Your hearing impairment: (1)Extremely severe (2)Severe (3)Moderate (4)Mild (5)Very mild (6)No restriction
s2.Your ear discharge: (1)Extremely severe (2)Severe (3)Moderate (4)Mild (5)Very mild (6)No restriction
s3.Your ear pain: (1)Extremely severe (2)Severe (3)Moderate (4)Mild (5)Very mild (6)No restriction
s4. The odor from your ear significantly bothers yourself and others (1)Absolutely true (2)True (3)Uncertain (4)False (5)Absolutely false |
s5.The hearing loss in your affected ear troubles you: (1)All of the time (2)Most of the time (3)A good bit of the time (4)Some of the time (5)A little of the time (6)None of the time
s6Over the past 6 months, please estimate how often your affected ear had purulent discharge: (1)Continuously (2)More than 5 times, but not continuously (3)3-4 times (4)1-2 times (5)Never
s7.The odor emanating from your affected ear significantly troubles you or others: (1)All of the time (2)Most of the time (3)A good bit of the time (4)Some of the time (5)A little of the time (6)None of the time
Medical Resource (MR) m1.How many times have you sought medical care for ear-related problems in the past 6 months? (1)More than 6 times (2)5-6 times (3)3-4 times (4)1-2 times (5)None
m2.Frequency of oral medication use for ear conditions during the past 6 months: (1)More than 6 times (2)5-6 times (3)3-4 times (4)1-2 times (5)None
m3.Frequency of topical medication use for ear conditions during the past 6 months: (1)More than 6 times (2)5-6 times (3)3-4 times (4)1-2 times (5)None
|
|
Note:Activity Restriction (AR): Limitations in daily activities caused by chronic otitis media with effusion.Symptoms (ST): Symptoms experienced by patients with chronic otitis media with effusion, including hearing loss and otorrhea.Medical Resource Utilization (MR): Frequency and details of medical visits for patients with chronic otitis media with effusion.Scoring:(1) = 1 point, (2) = 2 points, (3) = 3 points, (4) = 4 points, (5) = 5 points, (6) = 6 points.Higher total scores indicate better quality of life. |
|
The Chinese Version of the Chronic Ear Survey (CCES), as a validated quality-of-life assessment tool for otitis media, has been widely utilized in multiple clinical studies. For instance, Li Shan et al. applied this scale to compare postoperative quality of life in patients undergoing endoscopic versus microscopic tympanoplasty[8] , while Wu Peixia's research team employed it to evaluate the long-term efficacy and safety of balloon dilation eustachian tuboplasty combined with tympanostomy tube placement in patients with secretory otitis media[9] . Given its established reliability, validity, and extensive applicability in chronic secretory otitis media research, this study selected the CCES scale for systematic quality-of-life assessment in patients with CSOM.
Pharmacological Treatment Protocol
The study utilized triamcinolone acetonide nasal spray (55 μg per nostril, twice daily) with standardized administration techniques (pre-use shaking, lateral nasal wall spraying) and regular nasal mucosa monitoring during prolonged use, alongside eucalyptol/limonene/pinene enteric soft capsules (300 mg orally, three times daily) administered with cool water 30 minutes pre-meal, avoiding liquids above 40°C, and adjusted to postprandial dosing for gastrointestinal symptom mitigation.
Surgical Intervention Protocol
The tympanostomy procedure was performed under endoscopic guidance. Following satisfactory general anesthesia, the patient was positioned supine with the head rotated 30° toward the non-surgical ear. The operative field was disinfected and draped. A 0° rigid ear endoscope was introduced through the external auditory canal to visualize the tympanic membrane. Cerumen and secretions were meticulously cleared to optimize visualization. Anatomical landmarks (malleus handle and light reflex) were identified, with the incision site localized to the anteroinferior quadrant of the tympanic membrane. A 2-3 mm radial incision parallel to the tympanic annulus was created using a myringotomy knife. Middle ear effusions were aspirated using a microsuction device. A ventilation tube was inserted into the incision and secured via rotational manipulation. Residual blood and secretions were cleared from the external auditory canal, followed by endoscopic withdrawal after confirming hemostasis.
Statistical Methods
Patients completed questionnaires before and after treatment, with scores recorded according to the designated scale. Pre-treatment and post-treatment scores were analyzed using the two-sample t-test in SPSS 28.0. Pairwise comparisons were performed for score differences before and after different treatment modalities.
Results
Comparison of Baseline Characteristics Between the Two Groups
This study enrolled a total of 80 patients, with 38 in the myringotomy group and 42 in the pharmacotherapy group. All patients successfully completed treatment.No statistically significant differences were observed between the two groups in terms of age, sex, or education level (p > 0.05).(Table 2)
Table 2 Comparison of Baseline Characteristics Between Groups
|
Number of Cases (n) |
Gender Ratio (Male/Female) |
Mean Age (years) |
Educational Attainment Distribution (%) |
|||
|
|
Primary Education or Below |
Secondary Education (Junior/Senior High School) |
College Education or Above |
|||
|
Tympanostomy Group |
38 |
55.26/ 44.74 |
32.42 |
18.42 |
55.26 |
26.32 |
|
Pharmacotherapy Group |
42 |
57.14/ 42.86 |
31.75 |
16.67 |
59.52 |
23.81 |
This study enrolled a total of 80 patients, with 38 in the tympanostomy group and 42 in the pharmacotherapy group. All patients successfully completed treatment. No statistically significant differences were observed between the two groups in terms of age, gender, or educational level (p>0.05) (Table 2).
Comparison of Patients' Quality of Life Before and After Treatment
No statistically significant difference was observed between pre-treatment and post-treatment scores (P>0.05). Among the three subscales, no significant difference was found in the "Activity Limitation" scores before and after treatment. The "Symptom" scores showed significant improvement after treatment (P<0.05), indicating statistical significance. No statistically significant difference was noted in the "Healthcare Resource Utilization" scores before and after treatment (P>0.05).(Table 3)
Table 3 Comparison of Quality of Life Scores Before and After Treatment (Mean ± SD; n = 67)
|
|
Before Treatment (Score) |
After Treatment (Score) |
|
Activity Ristriction |
13.43±0.39 |
14.43±0.33 |
|
Symptom |
26.32±0.76 |
35.73±0.29 |
|
Medical Resource |
10.79±0.32 |
11.29±0.24 |
Comparison of Treatment Modalities on Quality of Life
Pairwise comparisons of score differences before and after treatment between tympanostomy tube insertion and pharmacotherapy showed no statistically significant differences in quality of life scores (pre- vs. post-treatment, P>0.05). Subgroup analyses revealed no significant variations in total scores between the two treatment modalities.(Table 4)
Table 4 Quality of Life Scores Before Treatment and 2-4 Months After Treatment Across Different Treatment Modalities (Mean ± SD; n = 67)
|
|
Before Treatment (Score) |
|
After Treatment 2-4months(Score) |
||||
|
|
Activity Ristriction |
Symptom |
Medical Resource |
|
Activity Ristriction |
Symptom |
Medical Resource |
|
Tympanostomy Group |
13.21±0.44 |
24.14±0.62 |
9.39±0.74 |
|
13.28±0.63 |
36.21±0.38 |
10.82±0.31 |
|
Pharmacotherapy Group |
13.62±0.23 |
26.47±0.58 |
10.21±0.36 |
|
14.91±0.28 |
35.15±0.82 |
11.15±0.53 |
Impact of Gender, Cost, and Education Level on Quality of Life
Univariate analysis of the three influencing factors—gender, cost, and education level—was conducted using t-test or ANOVA (F-test) (with homogeneity of variance confirmed by Levene's test). Results indicated no statistically significant correlation (P>0.05) between these factors and quality of life scores.
Discussion
Chronic Secretory Otitis Media (CSOM) represents a significant public health concern, with its detrimental effects on hearing impairment and quality of life having garnered widespread attention[10] . CSOM is characterized by the accumulation of fluid in the middle ear in the absence of acute infection signs. If left untreated, it may lead to long-term adverse consequences for auditory function and patient quality of life[11] .
Therefore, this study aimed to systematically evaluate the impact of different treatment modalities on quality of life and symptom management in CSOM patients[12] .Through a designed questionnaire, we compared the efficacy of surgical and pharmacological interventions. The results demonstrated significant improvement in symptom scores post-treatment; however, no statistically significant difference (p>0.05) was observed between the two treatment approaches regarding quality of life enhancement. These findings underscore the necessity for further investigation into the long-term outcomes of these interventions.
Our study not only enriches the existing body of knowledge but also highlights the importance of tailored treatment strategies for managing this prevalent condition[13] .The research addresses a critical gap in comparative effectiveness studies of CSOM treatments, particularly in evaluating differential impacts on patients' quality of life. Previous studies have predominantly focused on individual treatment outcomes without adequately comparing the relative efficacy of these interventions in improving overall quality of life[15] .
The results demonstrated that although symptom scores showed significant improvement, no statistically significant difference (p>0.05) was observed in quality-of-life scores between the tympanostomy tube insertion and pharmacological treatment groups. This suggests that both therapeutic approaches may provide comparable benefits, which aligns with previous studies and indicates no current consensus regarding the superiority of either treatment modality for CSOM. Further analysis revealed the complexity of patients' treatment experiences—while symptoms improved markedly, activity limitation scores showed no significant change, suggesting incomplete restoration of daily functioning. This discrepancy warrants further investigation into the physiological mechanisms of symptom relief and their clinical relevance to daily life.
Furthermore, future studies should investigate specific strategies to improve activity limitations, potentially incorporating patient-reported outcomes to better capture the multidimensional nature of quality of life in CSOM patients[16] . When analyzing demographic variables including gender, socioeconomic status, and educational background, we found these factors had no significant impact on quality-of-life outcomes. This finding emphasizes that treatment evaluation should focus on direct therapeutic effects rather than demographic characteristics. The absence of statistically significant differences across demographic groups suggests universal treatment responsiveness, which is crucial for clinicians when making treatment recommendations. Future research should explore cultural and psychosocial factors that may influence patients' treatment preferences and satisfaction, as these could significantly impact patient-centered CSOM management.
Several limitations of this study warrant careful consideration. A notable limitation is the relatively small sample size, which may compromise statistical power and the generalizability of the findings. Additionally, the study design did not incorporate laboratory assessments, potentially overlooking confounding variables that could influence treatment outcomes. The absence of long-term follow-up data also limits our ability to evaluate the durability of the observed therapeutic effects.
In summary, this study provides valuable insights into the impact of treatment modalities on quality of life in CSOM patients. The results demonstrate significant improvement in symptom scores post-treatment, though with limited effects on activity limitations. These findings underscore the necessity for ongoing evaluation of treatment strategies in otolaryngology to optimize patient care and refine therapeutic approaches. Future research should address the identified limitations to gain deeper understanding of treatment effects on long-term patient well-being.
Conclusion
This prospective comparison of surgical tympanostomy and pharmacotherapy in chronic secretory otitis media (CSOM) confirms both treatments significantly alleviate symptoms (e.g., hearing loss, tinnitus; p < 0.05) but fail to restore functional capacity or quality of life (QoL) (p > 0.05), with marginally greater symptom resolution in surgery. Persistent activity restrictions post-intervention highlight unaddressed functional limitations despite clinical improvements. Though QoL outcomes were unaffected by demographic factors, incomplete recovery necessitates personalized strategies integrating patient priorities. Study limitations (small n=80 cohort, short follow-up) mandate larger longitudinal trials to assess long-term efficacy, physiological mechanisms, and multidimensional QoL frameworks incorporating patient-reported outcomes.[1]
Abbreviations
|
Abbreviation |
Full Form |
|
CSOM |
Chronic Secretory Otitis Media |
|
QoL |
Quality of Life |
|
CCES |
Chinese Chronic Ear Survey |
|
ICD |
International Classification of Diseases |
|
AR |
Activity Restriction |
|
ST |
Symptom |
|
MR |
Medical Resource Utilization |
|
SPSS |
Statistical Package for the Social Sciences |
|
ANOVA |
Analysis of Variance |
Acknowledgements
The authors sincerely thank all participating patients and their families for their support and collaboration in this study.
Author contributions
TianLi Zhu and Min Luo conceived and designed the study and drafted the initial manuscript. Min Luo collected the data, and TianLi Zhu performed the data analysis. All authors critically reviewed the manuscript and approved the final version.
Ethics approval and consent to participate
This study did not require formal ethics committee approval due to its retrospective design and use of anonymized clinical data. All participants or their legal guardians provided written informed consent.
Competing Interests
The authors declare that they have no existing or potential commercial or financial relationships that could create a conflict of interest at the time of conducting this study.
Data Availability
All data needed to evaluate the conclusions in the paper are present in the paper or the Supplementary Materials. Additional data related to this paper may be requested from the authors.
References
[1] Rosenfeld RM, Shin JJ, Schwartz SR, Coggins R, Gagnon L, Hackell JM, et al. Clinical Practice Guideline: Otitis Media with Effusion Executive Summary (Update). Otolaryngol Head Neck Surg. 2016;154(2):201-214. https://doi.org/10.1177/0194599815624407.
[2] Lin W, Wang B, Wang M, Wang Y, Tuerdibieke Y, Qian C, et al. The Unneglectable Disease Burden of Otitis Media Among Children in China: A Systematic Review and Meta-analysis. Pediatr Infect Dis J. 2024;43(5):403-409. https://doi.org/10.1097/INF.0000000000004239.
[3] Clary RA, Bahadori RS, Muntz HR, Lusk RP. Bacteria in the middle ear and nasopharynx during tympanostomy tube insertion. Am J Otolaryngol. 1998 Sep-Oct;19(5):301-4. https://doi.org/10.1016/s0196-0709(98)90002-3.
[4] Chonmaitree T, Saeed K, Uchida T. A randomized, placebo-controlled trial of the effect of antihistamine or corticosteroid treatment in acute otitis media. J Pediatr. 2003;143(3):377-85. https://doi.org/10.1067/S0022-3476(03)00293-2.
[5] Yu X, Xu L, Xie Y, Huang M. Clinical study of the effect of mometasone furoate nasal spray treatment on hearing and in secretory otitis media in children. Clinics (Sao Paulo). 2024 Dec 12;80:100551. https://doi.org/10.1016/j.clinsp.2024.100551.
[6] Homøe P, Heidemann CH, Damoiseaux RA, Lailach S, Lieu JEC, Phillips JS, et al. Panel 5: Impact of otitis media on quality of life and development. Int J Pediatr Otorhinolaryngol. 2020 Mar;130 Suppl 1(Suppl 1):109837. https://doi.org/10.1016/j.ijporl.2019.109837.
[7] Nadol JB Jr, Staecker H, Gliklich RE. Outcomes assessment for chronic otitis media: the Chronic Ear Survey. Laryngoscope. 2000;110(3 Pt 3):32-35. https://doi.org/10.1097/00005537-200003002-00009.
[8] Li S, Ke J, Yang R, Zhang K, Pan T, Xin Y, et al. [Quality of life assessment after endoscopic and microscopic myringoplasty using Chinese version of the Zurich chronic middle ear inventory]. Lin Chuang Er Bi Yan Hou Tou Jing Wai Ke Za Zhi. 2021 Apr;35(4):297-301. Chinese. https://doi.org/10.13201/j.issn.2096-7993.2021.04.003.
[9] Wu P, Cao X, Zhang R, Liu Y, Li H, Wang W, et al. The long-term efficacy and safety of balloon dilation eustachian tuboplasty combined with tympanostomy tube insertion for patients with otitis media with effusion: study protocol for a prospective randomized controlled trial. Trials. 2024 Aug 29;25(1):572. https://doi.org/10.1186/s13063-024-08405-1.
[10] Magnan J. Otite séro-muqueuse [Secretory otitis media]. Rev Prat. 1998;48(8):860-865.
[11] Doyle KJ, Kong YY, Strobel K, Dallaire P, Ray RM. Neonatal middle ear effusion predicts chronic otitis media with effusion. Otol Neurotol. 2004;25(3):318-322. https://doi.org/10.1097/00129492-200405000-00020.
[12] Bhusal P, Harrison J, Sharma M, Jones DS, Hill AG, Svirskis D. Controlled release drug delivery systems to improve post-operative pharmacotherapy. Drug Deliv Transl Res. 2016;6(5):441-451. https://doi.org/10.1007/s13346-016-0305-z.
[13] Planchard D, Besse B. Vers l'individualisation des prescriptions en oncologie Cancer bronchique non à petites cellules [Towards customized therapy for non small cell lung cancer]. Bull Cancer. 2008;95(10):951-958. https://doi.org/10.1684/bdc.2008.0719.
[14] Zhao Z, Bai B, Wang S, et al. Physical and psychological correlates of somatic symptom in patients with functional constipation: a cross-sectional study. BMC Psychiatry. 2024;24(1):134. https://doi.org/10.1186/s12888-024-05559-9.
[15] Fakhry H, Goldenberg M, Sayer G, et al. Health-related quality of life in childhood cancer. J Dev Behav Pediatr. 2013;34(6):419-440. https://doi.org/10.1097/DBP.0b013e31828c5fa6.
[16] Church J. Quality of life and patient-reported outcomes. Br J Surg. 2018;105(3):157-158. https://doi.org/10.1002/bjs.10824.
 Figures
Figures References
References Peer
Peer Information
Information[1] Rosenfeld RM, Shin JJ, Schwartz SR, Coggins R, Gagnon L, Hackell JM, et al. Clinical Practice Guideline: Otitis Media with Effusion Executive Summary (Update). Otolaryngol Head Neck Surg. 2016;154(2):201-214. https://doi.org/10.1177/0194599815624407.
[2] Lin W, Wang B, Wang M, Wang Y, Tuerdibieke Y, Qian C, et al. The Unneglectable Disease Burden of Otitis Media Among Children in China: A Systematic Review and Meta-analysis. Pediatr Infect Dis J. 2024;43(5):403-409. https://doi.org/10.1097/INF.0000000000004239.
[3] Clary RA, Bahadori RS, Muntz HR, Lusk RP. Bacteria in the middle ear and nasopharynx during tympanostomy tube insertion. Am J Otolaryngol. 1998 Sep-Oct;19(5):301-4. https://doi.org/10.1016/s0196-0709(98)90002-3.
[4] Chonmaitree T, Saeed K, Uchida T. A randomized, placebo-controlled trial of the effect of antihistamine or corticosteroid treatment in acute otitis media. J Pediatr. 2003;143(3):377-85. https://doi.org/10.1067/S0022-3476(03)00293-2.
[5] Yu X, Xu L, Xie Y, Huang M. Clinical study of the effect of mometasone furoate nasal spray treatment on hearing and in secretory otitis media in children. Clinics (Sao Paulo). 2024 Dec 12;80:100551. https://doi.org/10.1016/j.clinsp.2024.100551.
[6] Homøe P, Heidemann CH, Damoiseaux RA, Lailach S, Lieu JEC, Phillips JS, et al. Panel 5: Impact of otitis media on quality of life and development. Int J Pediatr Otorhinolaryngol. 2020 Mar;130 Suppl 1(Suppl 1):109837. https://doi.org/10.1016/j.ijporl.2019.109837.
[7] Nadol JB Jr, Staecker H, Gliklich RE. Outcomes assessment for chronic otitis media: the Chronic Ear Survey. Laryngoscope. 2000;110(3 Pt 3):32-35. https://doi.org/10.1097/00005537-200003002-00009.
[8] Li S, Ke J, Yang R, Zhang K, Pan T, Xin Y, et al. [Quality of life assessment after endoscopic and microscopic myringoplasty using Chinese version of the Zurich chronic middle ear inventory]. Lin Chuang Er Bi Yan Hou Tou Jing Wai Ke Za Zhi. 2021 Apr;35(4):297-301. Chinese. https://doi.org/10.13201/j.issn.2096-7993.2021.04.003.
[9] Wu P, Cao X, Zhang R, Liu Y, Li H, Wang W, et al. The long-term efficacy and safety of balloon dilation eustachian tuboplasty combined with tympanostomy tube insertion for patients with otitis media with effusion: study protocol for a prospective randomized controlled trial. Trials. 2024 Aug 29;25(1):572. https://doi.org/10.1186/s13063-024-08405-1.
[10] Magnan J. Otite séro-muqueuse [Secretory otitis media]. Rev Prat. 1998;48(8):860-865.
[11] Doyle KJ, Kong YY, Strobel K, Dallaire P, Ray RM. Neonatal middle ear effusion predicts chronic otitis media with effusion. Otol Neurotol. 2004;25(3):318-322. https://doi.org/10.1097/00129492-200405000-00020.
[12] Bhusal P, Harrison J, Sharma M, Jones DS, Hill AG, Svirskis D. Controlled release drug delivery systems to improve post-operative pharmacotherapy. Drug Deliv Transl Res. 2016;6(5):441-451. https://doi.org/10.1007/s13346-016-0305-z.
[13] Planchard D, Besse B. Vers l'individualisation des prescriptions en oncologie Cancer bronchique non à petites cellules [Towards customized therapy for non small cell lung cancer]. Bull Cancer. 2008;95(10):951-958. https://doi.org/10.1684/bdc.2008.0719.
[14] Zhao Z, Bai B, Wang S, et al. Physical and psychological correlates of somatic symptom in patients with functional constipation: a cross-sectional study. BMC Psychiatry. 2024;24(1):134. https://doi.org/10.1186/s12888-024-05559-9.
[15] Fakhry H, Goldenberg M, Sayer G, et al. Health-related quality of life in childhood cancer. J Dev Behav Pediatr. 2013;34(6):419-440. https://doi.org/10.1097/DBP.0b013e31828c5fa6.
[16] Church J. Quality of life and patient-reported outcomes. Br J Surg. 2018;105(3):157-158. https://doi.org/10.1002/bjs.10824.
Peer-review Terminology
Identity transparency: Single anonymized
Reviewer interacts with: Editor
Details
This is an open access article under the terms of the Creative Commons Attribution License(http://creativecommons.org/licenses/by/4.0/), which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Publication History
Received 2025-04-15
Accepted 2025-05-17
Published 2025-05-19


