Abstract
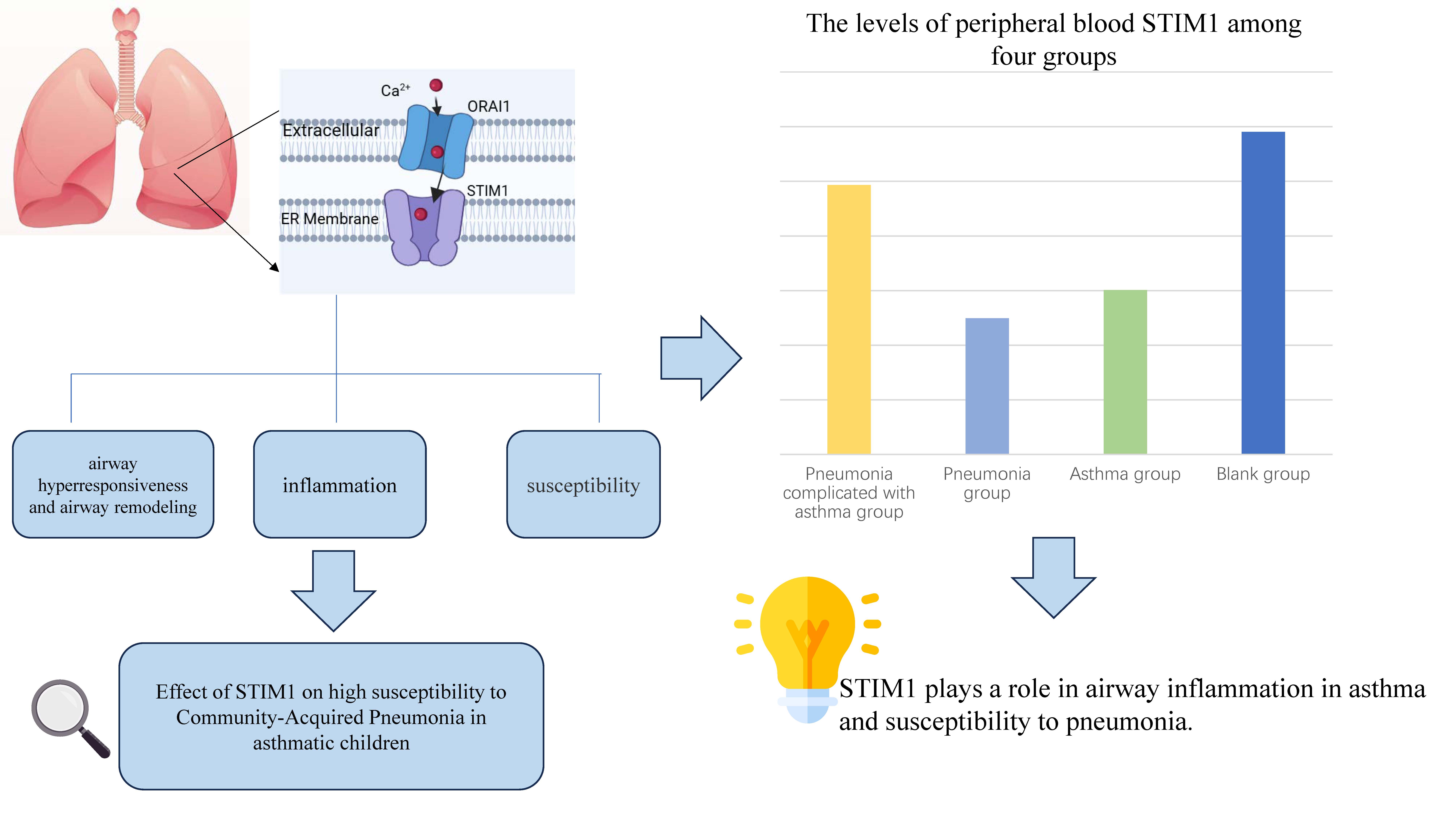
Aim:To study the correlation between serum STIM1 level and high susceptibility to pneumonia in children with asthma.
Methods:A total of 116 children hospitalized at our facility as the study subjects from March 2023 to February 2024, according to the including and exclusion criteria, which were categorized into three groups: pneumonia complicated with asthma group, pneumonia group, asthma group. 30 healthy children were selected as a blank group. The concentrations of serum STIM1 were measured by ELISA and compared across all groups.
Results: The study measured serum STIM1 concentrations (pg/ml) in the four groups, respectively. The data showed that there were statistical differences in serum STIM1 concentration in different conditions (P < 0.05). Subsequent multiple comparisons revealed that the serum STIM1 concentration in the pneumonia complicated with asthma group was significantly higher than that in both the pneumonia group (P < 0.001) and the asthma group (P < 0.001). Additionally, serum STIM1 concentrations in the pneumonia complicated with asthma group (P < 0.05), pneumonia group (P < 0.001), and no statistically significant difference was found between the asthma group and the pneumonia group (P > 0.05).
Conclusions:STIM1 plays a role in airway inflammation in asthma and susceptibility to pneumonia. Targeting this pathway may provide novel therapeutic strategies for high-risk children with asthma complicated by pneumonia, though further validation of its safety and efficacy is required.
Keywords:Asthma; Pneumonia; STIM1; Susceptibility; Children
Introduction
Bronchial asthma is a chronic inflammatory respiratory disease characterized by recurrent bronchospasms triggered by respiratory allergies and other factors. This condition involves inflammation and narrowing of the airways, leading to symptoms such as wheezing, shortness of breath, chest tightness, and coughing. The primary pathophysiological features of asthma include airway hyperresponsiveness (AHR) and airway remodeling (AR) [1~2] . In China, pneumonia is the leading cause of death in children under 5 years of age, with the majority of these fatalities resulting from community-acquired pneumonia (CAP) [3] . Asthma is closely associated with pneumonia, as respiratory infections can precipitate acute asthma attacks. Children with asthma are at an increased risk of respiratory infections, which may subsequently lead to the development of CAP, including infections caused by viruses, bacteria, fungi and atypical pathogens [4] . However, a detailed and comprehensive explanation of the specific mechanisms underlying this correlation remains lacking.
Stromal interaction molecule 1 (STIM1), as a calcium ion receptor, and calcium release-activated calcium modulator 1(ORAI1), as a calcium ion channel, are key proteins involved in the transmembrane transport of calcium ions and the transduction of calcium ion signals, which have important physiological functions. Martin et al. found that STIM1 was involved in the contraction and proliferation of airway smooth muscle (ASM) and was the core initiating factor of AHR and AR. [4] It was observed that STIM1 expression was upregulated in the AS, of asthmatic mice by constructing a chronic asthma mouse model and manipulating the expression of STIM1 in ASM. STIM1 is essential for the metabolic and transcriptional reprograming of ASM, which includes ASM proliferation, cytokine and extracellular matrix secretion, increased mitochondrial mass, and enhanced oxidative phosphorylation and glycolytic flux. Furthermore, STIM1 contributes to AHR in asthmatic mice by increasing both the frequency and amplitude of intracytoplasmic calcium oscillations in ASM. Many studies have shown that STIM1 plays an important role in inducing alveolar epithelial cell injury, including viral infection, bacterial infection, poisoning and ventilator-related lung injury [5~6] . One of the involved mechanisms is that in these lung injuries, STIM1 expression is up-regulated, calcium ion influx is overloaded and endothelial permeability is increased. STIM1 plays an important role in asthma and lung damage. Therefore, it is hypothesized whether STIM1 plays an important role in the mechanism of asthma patients being highly susceptible to pneumonia. Based on these findings, This study enrolled 146 pediatric subjects, selected 30 healthy children from the 146 participants, aiming to explore the correlation between serum STIM1 level and the high susceptibility of asthmatic children to CAP, and provide new ideas for further exploring the mechanism of this high susceptibility.
Materials and Methods
General information
This study is a prospective cohort trial. Following the established inclusion and exclusion criteria, a total of 116 children were selected as study subjects after receiving approval from the hospital's ethics committee. The children were categorized into three groups: pneumonia complicated with asthma group, asthma group and pneumonia group. Concurrently, 30 healthy children were selected to serve as a blank group, from which venous blood samples were collected.
Inclusion criteria
According to China's guidelines on community-based pneumonia and asthma in children published in 2019 and 2020, children aged 3 to 14 who meet the relevant diagnostic criteria [1,3] , regardless of gender, are included in the following groups:
(1) Pneumonia complicated with asthma group: Clinically diagnosed with bronchial asthma, receiving regular treatment, in the chronic persistent phase of asthma, with no acute asthma attacks during hospitalization; hospitalized for community-acquired pneumonia within 3-5 days of the disease course.(2) Asthma Group: Clinically diagnosed with bronchial asthma, receiving regular treatment, in the chronic persistent phase of asthma, hospitalized for acute asthma exacerbation without infection as a triggering factor.(3) Pneumonia Group: Hospitalized for community-acquired pneumonia within 3-5 days of the disease course, with non-severe pneumonia.(4) Blank Group: Meets the diagnostic criteria for familial short stature and has had no respiratory infections within the past 4 weeks.
Exclusion Criteria
(1) Presence of other underlying diseases such as congenital heart disease, congenital lung developmental disorders, immune dysfunction, or immunodeficiency;(2) Refusal to undergo blood testing;(3) Use of glucocorticoids and/or immunosuppressants within 4 weeks prior to specimen collection;Incomplete medical records.
Sample collection and testing
A volume of 2 mL of fasting venous blood was collected from all subjects and placed in a separation glue-coagulant tube. The samples were allowed to stand at room temperature for 30 minutes before being centrifuged at 2500 rpm for 15 minutes. The upper serum was collected and aliquoted into small EP tubes, which were subsequently frozen and stored at -80°C for future use. The serum levels of STIM1 and ORAI1 were measured using Enzyme-Linked Immunosorbent Assay (ELISA), and the STIM1 assay kit was purchased from Shanghai Enzyme-linked Biotechnology Co., Ltd. The optical density (OD) values were detected at a wavelength of 450 nm using a microplate reader, and the concentrations of target markers in the samples were calculated by plotting standard curves , and detailed procedures followed the kit manufacturer's guidelines.
Statistical analysis
GraphPad Prism 10 was utilized for statistical analysis. Categorical data were represented as "n " and analyzed using the chi-square test. Continuous data were expressed as mean standard deviation (X ± SD). One-way analysis of variance was employed for multi-group comparisons, and the Bonferroni test was applied for pairwise comparisons. In this study, P < 0.05 was considered statistically significant.
Results
General information
A total of 146 hospitalized children were included in this study. All data adhered to a normal distribution. No significant differences were observed in gender, age, and weight across all groups (P > 0.05) (Table 1).
Table 1. Comparison of baseline date
|
Index |
|
Pneumonia complicated with asthma group (n=40) |
Pneumonia group (n=42) |
Asthma group (n=34) |
Blank group. (n=30) |
P-value |
|
Age (year) |
|
5.63±2.55 |
5.36±2.17 |
5.68±2.70 |
6.43±2.55 |
0.33 |
|
Weight (kg) |
|
23.5±10.40 |
21.65±8.35 |
24.71±11.00 |
24.13±10.48 |
0.57 |
|
Gender [n] |
Male |
19 |
21 |
19 |
16 |
0.99 |
|
|
Female |
21 |
21 |
15 |
14 |
Serum STIM1 levels
Serum STIM1 concentration were measured in the pneumonia complicated with asthma group, the pneumonia group, the asthma group and the blank group. The data indicated statistically significant differences in serum STIM1 concentration across the different disease states (P < 0.05). Subsequent multiple comparisons revealed that the serum STIM1 concentration in the pneumonia complicated with asthma group was significantly higher than that in both the pneumonia group (P < 0.001) and the asthma group (P < 0.001). Additionally, serum STIM1 concentrations in the pneumonia complicated with asthma group (P < 0.05), pneumonia group (P < 0.001), and no statistically significant difference was found between the asthma group and the pneumonia group (P > 0.05) (Table 2 and Figure 1).
Figure 1 indicates that there are differences in serum STIM1 concentrations among the groups. The asthma group and the pneumonia group showed significantly lower STIM1 concentrations compared to the blank group. The pneumonia complicated with asthma group exhibited higher STIM1 concentrations compared to the pneumonia group and the asthma group, but the levels were still lower than those in the blank group.
Table 2. The levels of peripheral blood STIM1 among pneumonia complicated with asthma group, pneumonia group, asthma group and blank group.
|
Index |
Pneumonia complicated with asthma group |
Pneumonia group |
Asthma group |
Blank group |
F-value |
P-value |
|
STIM1 (X̅ ± SD) (pg/ml) |
398.6±17.54b,** |
349.9±17.84a,b,*** |
360.2±35.88a,b,*** |
418.1±14.40a,*** |
69.93 |
<0.05 |
‘a’ stands for statistically significant compared to pneumonia complicated with asthma group (P<0.05); ‘b’ stands for statistically significant compared to blank group (P<0.05); ‘**’ stands for P-value is less than 0.05; ‘***’ stands for P-value is less than 0.001.
Discussion
Asthma, the most common chronic respiratory disease in children, is characterized by chronic airway inflammation and bronchial hyperreactivity. Pneumonia, particularly community-acquired pneumonia (CAP), is a leading cause of death in children under five in China. Asthma and CAP are closely linked—respiratory infections can trigger asthma exacerbations, while asthmatic children are more susceptible to infections. However, the underlying mechanisms remain unclear.
Calcium (Ca²⁺) signaling, mediated by STIM1 (endoplasmic reticulum Ca²⁺ sensor) and ORAI1 (plasma membrane Ca²⁺ channel), plays a key role in airway smooth muscle (ASM) contraction, immune cell function, and inflammation. Store-operated Ca²⁺ entry (SOCE) regulates intracellular Ca²⁺ homeostasis and is critical in asthma pathogenesis:(1) ASM remodeling: STIM1 upregulation enhances Ca²⁺ signaling, promoting ASM proliferation and airway hyperresponsiveness (AHR). (2) Immune modulation: STIM1/ORAI1 are essential for T-cell activation and immune tolerance. Their dysfunction can lead to immunodeficiency or autoimmunity [7~14] . (3) Antiviral response: STIM1/ORAI1 regulate baseline IFN-β levels, influencing host resistance to viral infections (e.g., SARS-CoV-2) [15,16] .Targeting STIM1/SOCE may alleviate AHR and airway remodeling (AR) in asthma. For example, a short palate lung and nasal epithelial clone1 (SPLUNC1) peptide mimetics inhibit SOCE, reducing lung inflammation [6] . Additionally, STIM1 deficiency in ASM or immune cells alters disease progression, highlighting its therapeutic potential [5] . In summary, STIM1/ORAI1-mediated Ca²⁺ signaling links asthma pathophysiology (AHR, AR, and inflammation) with pneumonia susceptibility, offering novel intervention strategies.
The results of this study indicate that STIM1 is involved in the pathological processes of asthma and play a negative role in its pathogenesis. However, the result obtained in this study does not fully align with this expectation. As blood samples were collected for this experiment, the potential impact of the numerous immune and inflammatory cells present in the blood on serum STIM1 levels warrants further investigation. As immunological research progresses, an increasing number of studies have unveiled the complex immunological phenomena associated with asthma. In children with asthma, immune function is disturbed. It characterized by a decreased CD4+/CD8+ T cell ratio and an increased Th17/Treg ratio, both of which are significant immunological features. [18] Studies have shown that STIM1 plays a crucial role in the proliferation and differentiation of immune cells, while SOCE regulates the anabolic growth of T lymphocytes by enhancing glucose uptake. Glucose transporter protein 1 (GLUT1) as an important kind of membrane protein found both intracellularly and extracellularly, is essential for glucose transport. Its unique structure and function provide cells with the necessary energy source, maintaining intracellular energy balance and survival, which is particularly important for activated CD4+ T cells in glucose uptake. In CD8+ T cells, a deficiency in SOCE partially affects the expression of GLUT1 protein. Notably, STIM1 exhibits a greater capacity to promote the anabolic growth of CD4+ T cell than CD8+ T cell [18] . In the study of Martin et al. [5] , it was found that STIM1 is involved in regulating the process of glycolysis. In the STIM1 knockout mouse model, glucose consumption and lactate production were reduced, and the expression of ASM GLUT1 was reduced. STIM1 has the effect of promoting the metabolism and synthesis of CD4+T cells, and its effect is greater than that of CD8+T cells. This conclusion is consistent with the results of this experiment. It suggests that STIM1 is involved in one of the important pathogenic mechanisms in the occurrence and development of asthma, namely Immune imbalance. Concurrently, research has identified that microRNA (miRNA) plays a vital role in regulating the balance between Th17 and Treg cells; specifically, the overexpression of microRNA 641 (miR-641) may downregulate its target genes STIM1 and Special AT-rich Sequence Binding Protein 1 (SATB1), thereby inducing Th17 (upregulated) /Treg (downregulated) imbalance [19] . That is, when the production of STIM1 is inhibited, Th17 is up-regulated, Treg is down-regulated, and the Th17/Treg ratio increases. In asthma patients, the Th17/Treg ratio is up-regulated, [17] , so it is consistent with the experimental results. The results of this experiment indicated that the level of serum STIM1 in asthmatic children was reduced, aligning with previous observations. In summary, the decreased expression level of serum STIM1 in children with asthma may be related to their immune imbalance, but the specific mechanism still needs further study.
Numerous studies have highlighted the significant role of STIM1 in the inflammatory injury of alveolar epithelial cells. Research indicates that serum STIM1 is highly expressed in patients infected with swine flu, and the STIM1/miR-223/NLRP3 axis regulates the inflammation of lung epithelial cells induced by the influenza A virus. The NLRP3 inflammasome serves as a central recognition sensor for cellular stress as well as for extracellular viral and bacterial infections. Silencing STIM1 can enhance the expression of miR-223 and inactivate the NLRP3 inflammasome, thereby mitigating the inflammatory damage caused by the influenza A virus to lung epithelial cells [20] . Additionally, streptococcal hemolysin O (SLO), a cholesterol-dependent cellular hemolysin derived from group A streptococcus, induces various types of Ca2+ signaling in host cells and plays a key role in membrane repair and cell fate determination. SLO-mediated Ca2+ signaling involves Ca2+ release from intracellular stores and SOCE. The phenotype of Ca2+ signaling is influenced by the expression levels of STIM1 and ORAI1, suggesting that STIM1 is pivotal in streptococcus-induced lung infections and pneumonia [21] . Studies have demonstrated that paraquat (PQ) acts as an agonist of STIM1, leading to increased intracellular calcium levels. STIM1 is crucial for inducing cell cycle arrest and cell death in PQ-mediated acute lung injury [22~23] . Concurrently, ventilator-induced lung injury is characterized by heightened endothelial permeability, which can be modulated by SOCE. Research involving related mouse models has revealed that STIM1 and ORAI1 expressions are upregulated in response to high tidal volume exposure. This upregulation on results in calcium overload and increased endothelial permeability, ultimately contributing to lung injury [25] .
Children with CAP exhibit multifaceted cellular immune dysfunction, primarily characterized by:(1) T lymphocyte subset imbalance: Ⅰ: CD4+ T cells: Disrupted Th1/Th2 balance (Th1 responses [anti-viral/intracellularimmunity] are suppressed, while Th2 responses [allergy/anti-parasiticimmunity] are upregulated);Ⅱ:CD8+ T cells: Impaired cytotoxic function, reducing infected cell clearance; Ⅲ:Treg/Th17 imbalance: Decreased regulatory T cells (Treg) and increased pro-inflammatory Th17 cells, promoting excessive inflammation and tissue damage;(2) NK cell activity dysregulation;(3) Macrophage phagocytic dysfunction;(4) Cytokine network disruption [26] .
The results of this experiment indicated that the serum STIM1 concentration in the pneumonia group was significantly lower than that in the control group. This suggests that STIM1 may play a negative role in infectious injury. Additionally, the study demonstrated statistically significant differences in serum STIM1 concentrations across various disease states. Further analysis revealed that the serum STIM1 concentration in children with asthma complicated with pneumonia was significantly lower than that of the blank group, yet significantly higher than that of both the pneumonia and asthma groups. Additional assessments indicated that STIM1 negatively influenced the occurrence and progression of asthma and pneumonia, with a similar negative effect observed in cases of coexisting asthma and pneumonia; however, the degree of this negative effect did not increase significantly, and appeared to trend downward. Numerous studies have established a connection between asthma and pneumonia, yet the precise mechanism remains unclear. Based on the current research findings, we hypothesize that serum STIM1 concentrations in children with pneumonia should be significantly elevated, and a positive correlation between these two markers is expected. Moreover, the levels of STIM1 should be markedly higher in patients with asthma and pneumonia, with serum STIM1 concentrations anticipated to further increase in children suffering from both conditions.
Regrettably, there is currently no research investigating the role of STIM1 in the mechanism underlying asthma's susceptibility to pneumonia. The results of this experiment showed that serum STIM1 concentration in the asthma and pneumonia group was significantly higher than that in the asthma and pneumonia groups, with no significant difference observed between the asthma and pneumonia groups. It is suggested that STIM1 plays a role in the high susceptibility to pneumonia in asthma, consistent with the anticipated results partly. Although the outcomes of this experiment did not entirely align with expectations, they nonetheless indicate that STIM1 plays significant roles in the disease process associated with asthma-related pneumonia susceptibility. This finding offers new insights and guidance for further investigation into the pathophysiological mechanisms underlying asthma high susceptibility to pneumonia.
The role of STIM1 in the mechanism underlying the heightened susceptibility to pneumonia in asthma has not been previously investigated, marking the innovation of this study. However, certain limitations exist within this research. The protein activity indices of STIM1 before and after treatment were not assessed, resulting in a lack of dynamic change data. For ethical reasons, the levels of STIM1 in the bronchoalveolar lavage fluid of the study subjects were not measured, and airway epithelial tissue samples were not collected for protein analysis. Furthermore, the impact of various immune cell populations in the blood on serum levels of STIM1 was still unclear, indicating a need for a more comprehensive experimental design. This study was conducted at a single center with a limited sample size, necessitating multi-center studies with larger cohorts to better elucidate the roles of STIM1 in this increased susceptibility.
Conclusion
In summary, STIM1 is significant contributors to the increased susceptibility of asthma to pneumonia, providing a foundation for further exploration of the mechanisms involved, with the aim of offering more accurate references for clinical diagnosis and treatment.
Acknowledgements
Not applicable.
Author contributions
Bei-bei Yang: Conceptualization, data curation, investigation, methodology, project administration, software, resources, writing–original draft writing; Yu-huan Deng: data curation, investigation; Sheng-gang Ding: Supervision, validation, funding acquisition, writing–review & editing. All authors have read and agreed to the published version of the manuscript.
Ethics approval and consent to participate
Informed consent was obtained from the relatives of the participating children, and the clinical data will be analyzed while ensuring the anonymity of the children's identities. The experimental procedures outlined in this study have been reviewed and approved by the Clinical Research Ethics Committee of the First Affiliated Hospital of Anhui Medical University (No. PJ 2024-03-24). These procedures comply with the ethical standards for medical research involving human subjects as established in the 1964 Declaration of Helsinki and its subsequent amendments.
Funding information
This work was supported by General Program of National Natural Science Foundation of China (Grant numbers 52273113).
Competing Interests
The authors declare that they have no existing or potential commercial or financial relationships that could create a conflict of interest at the time of conducting this study.
Data Availability
All data needed to evaluate the conclusions in the paper are present in the paper or the Supplementary Materials. Additional data related to this paper may be requested from the authors.
References
[1].Asthma group of Chinese Throacic Society (2020). Zhonghua jie he he hu xi za zhi = Zhonghua jiehe he huxi zazhi = Chinese journal of tuberculosis and respiratory diseases, 43(12), 1023–1048. https://doi.org/10.3760/cma.j.cn112147-20200618-00721.
[2].Huang K, Yang T, Xu J, et al. Prevalence, risk factors, and management of asthma in China: a national cross-sectional study. Lancet. 2019;394(10196):407-418. https://doi.org/10.1016/S0140-6736(19)31147-X.
[3].National Health Commission of the People’s Republic of China, State Administration of Traditional Chinese Medicine. Guideline for diagnosis and treatment of community-acquired pneumonia in Children (2019 version). Chin J Clin Infect Dis, 2019, 12(1): 6-13. https://doi.org/ 10.3760/cma.j.issn.1674-2397.2019.01.002.
[4].Xepapadaki P, Koutsoumpari I, Papaevagelou V, Karagianni C, Papadopoulos NG. Atypical bacteria and macrolides in asthma. Allergy Asthma Clin Immunol. 2008;4(3):111-116. https://doi.org/10.1186/1710-1492-4-3-111.
[5].Johnson MT, Xin P, Benson JC, et al. STIM1 is a core trigger of airway smooth muscle remodeling and hyperresponsiveness in asthma. Proc Natl Acad Sci U S A. 2022;119(1):e2114557118. https://doi.org/10.1073/pnas.2114557118.
[6].Wrennall JA, Ahmad S, Worthington EN, et al. A SPLUNC1 Peptidomimetic Inhibits Orai1 and Reduces Inflammation in a Murine Allergic Asthma Model. Am J Respir Cell Mol Biol. 2022;66(3):271-282. https://doi.org/10.1165/rcmb.2020-0452OC.
[7].Srikanth S, and Gwack Y. 2013. Orai1-NFAT signalling pathway triggered by T cell receptor stimulation. Mol Cells 35: 182–194.
[8].Desvignes L, Weidinger C, Shaw P, et al. STIM1 controls T cell-mediated immune regulation and inflammation in chronic infection. J Clin Invest. 2015;125(6):2347-2362. https://doi.org/10.1172/JCI80273
[9].Prakriya M, Lewis RS. Store-Operated Calcium Channels. Physiol Rev. 2015;95(4):1383-1436. https://doi.org/10.1152/physrev.00020.2014.
[10].Gwack Y, Srikanth S, Oh-Hora M, et al. Hair loss and defective T- and B-cell function in mice lacking ORAI1. Mol Cell Biol. 2008;28(17):5209-5222. https://doi.org/10.1128/MCB.00360-08;
[11].Vig M, DeHaven WI, Bird GS, et al. Defective mast cell effector functions in mice lacking the CRACM1 pore subunit of store-operated calcium release-activated calcium channels. Nat Immunol. 2008;9(1):89-96. https://doi.org/10.1038/ni1550.
[12].Oh-Hora M, Yamashita M, Hogan PG, et al. Dual functions for the endoplasmic reticulum calcium sensors STIM1 and STIM2 in T cell activation and tolerance. Nat Immunol. 2008;9(4):432-443. https://doi.org/10.1038/ni1574.
[13].Qu B, Al-Ansary D, Kummerow C, Hoth M, Schwarz EC. ORAI-mediated calcium influx in T cell proliferation, apoptosis and tolerance. Cell Calcium. 2011;50(3):261-269. https://doi.org/10.1016/j.ceca.2011.05.015.
[14].Matsumoto M, Fujii Y, Baba A, Hikida M, Kurosaki T, Baba Y. The calcium sensors STIM1 and STIM2 control B cell regulatory function through interleukin-10 production. Immunity. 2011;34(5):703-714. https://doi.org/10.1016/j.immuni.2011.03.016.
[15].Wu B, Ramaiah A, Garcia G Jr, Hasiakos S, Arumugaswami V, Srikanth S. ORAI1 Limits SARS-CoV-2 Infection by Regulating Tonic Type I IFN Signaling. J Immunol. 2022;208(1):74-84. https://doi.org/10.4049/jimmunol.2100742.
[16].Srikanth S, Woo JS, Wu B, et al. The Ca2+ sensor STIM1 regulates the type I interferon response by retaining the signaling adaptor STING at the endoplasmic reticulum. Nat Immunol. 2019;20(2):152-162. https://doi.org/10.1038/s41590-018-0287-8.
[17].Bryant N, Muehling LM. T-cell responses in asthma exacerbations. Ann Allergy Asthma Immunol. 2022;129(6):709-718. https://doi.org/10.1016/j.anai.2022.07.027.
[18].Vaeth M, Maus M, Klein-Hessling S, et al. Store-Operated Ca2+ Entry Controls Clonal Expansion of T Cells through Metabolic Reprogramming. Immunity. 2017;47(4):664-679.e6. https://doi.org/10.1016/j.immuni.2017.09.003.
[19].Zhu H, Ruan X, Zhao K, et al. The miR-641-STIM1 and SATB1 axes play important roles in the regulation of the Th17/Treg balance in ITP. Sci Rep. 2024;14(1):11243. Published 2024 May 16. https://doi.org/10.1038/s41598-024-61660-9.
[20].Liu CC, Miao Y, Chen RL, et al. STIM1 mediates IAV-induced inflammation of lung epithelial cells by regulating NLRP3 and inflammasome activation via targeting miR-223. Life Sci. 2021;266:118845. https://doi.org/10.1016/j.lfs.2020.118845.
[21].Usmani SM, von Einem J, Frick M, et al. Molecular basis of early epithelial response to streptococcal exotoxin: role of STIM1 and Orai1 proteins. Cell Microbiol. 2012;14(3):299-315. https://doi.org/10.1111/j.1462-5822.2011.01724.x.
[22].Fan H, Huang H, Hu L, et al. The activation of STIM1 mediates S-phase arrest and cell death in paraquat induced acute lung intoxication. Toxicol Lett. 2018;292:123-135. https://doi.org/10.1016/j.toxlet.2018.04.030.
[23].Yang W, Tian R, Zhu Y, et al. Paraquat is an agonist of STIM1 and increases intracellular calcium levels. Commun Biol. 2022;5(1):1151. Published 2022 Oct 30. https://doi.org/10.1038/s42003-022-04130-0.
[24].Song X, Liu Y, Dong L, Wang Y. Stromal-Interacting Molecule 1 (Stim1)/Orai1 Modulates Endothelial Permeability in Ventilator-Induced Lung Injury. Med Sci Monit. 2018;24:9413-9423. Published 2018 Dec 27. https://doi.org/10.12659/MSM.911268.
[25].Yuan XH, Li YM, Shen YY, Yang J, Jin Y. Clinical and Th1/Th2 immune response features of hospitalized children with human rhinovirus infection. J Med Virol. 2020;92(1):26-33. https://doi.org/10.1002/jmv.25587.
[26].Thomas R, Qiao S, Yang X. Th17/Treg Imbalance: Implications in Lung Inflammatory Diseases. Int J Mol Sci. 2023;24(5):4865. Published 2023 Mar 2. https://doi.org/10.3390/ijms24054865.
 Figures
Figures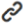 References
References Peer
Peer Information
InformationFigure 1 indicates that there are differences in serum STIM1 concentrations among the groups. The asthma group and the pneumonia group showed significantly lower STIM1 concentrations compared to the blank group. The pneumonia complicated with asthma group exhibited higher STIM1 concentrations compared to the pneumonia group and the asthma group, but the levels were still lower than those in the blank group.

[1].Asthma group of Chinese Throacic Society (2020). Zhonghua jie he he hu xi za zhi = Zhonghua jiehe he huxi zazhi = Chinese journal of tuberculosis and respiratory diseases, 43(12), 1023–1048. https://doi.org/10.3760/cma.j.cn112147-20200618-00721.
[2].Huang K, Yang T, Xu J, et al. Prevalence, risk factors, and management of asthma in China: a national cross-sectional study. Lancet. 2019;394(10196):407-418. https://doi.org/10.1016/S0140-6736(19)31147-X.
[3].National Health Commission of the People’s Republic of China, State Administration of Traditional Chinese Medicine. Guideline for diagnosis and treatment of community-acquired pneumonia in Children (2019 version). Chin J Clin Infect Dis, 2019, 12(1): 6-13. https://doi.org/ 10.3760/cma.j.issn.1674-2397.2019.01.002.
[4].Xepapadaki P, Koutsoumpari I, Papaevagelou V, Karagianni C, Papadopoulos NG. Atypical bacteria and macrolides in asthma. Allergy Asthma Clin Immunol. 2008;4(3):111-116. https://doi.org/10.1186/1710-1492-4-3-111.
[5].Johnson MT, Xin P, Benson JC, et al. STIM1 is a core trigger of airway smooth muscle remodeling and hyperresponsiveness in asthma. Proc Natl Acad Sci U S A. 2022;119(1):e2114557118. https://doi.org/10.1073/pnas.2114557118.
[6].Wrennall JA, Ahmad S, Worthington EN, et al. A SPLUNC1 Peptidomimetic Inhibits Orai1 and Reduces Inflammation in a Murine Allergic Asthma Model. Am J Respir Cell Mol Biol. 2022;66(3):271-282. https://doi.org/10.1165/rcmb.2020-0452OC.
[7].Srikanth S, and Gwack Y. 2013. Orai1-NFAT signalling pathway triggered by T cell receptor stimulation. Mol Cells 35: 182–194.
[8].Desvignes L, Weidinger C, Shaw P, et al. STIM1 controls T cell-mediated immune regulation and inflammation in chronic infection. J Clin Invest. 2015;125(6):2347-2362. https://doi.org/10.1172/JCI80273
[9].Prakriya M, Lewis RS. Store-Operated Calcium Channels. Physiol Rev. 2015;95(4):1383-1436. https://doi.org/10.1152/physrev.00020.2014.
[10].Gwack Y, Srikanth S, Oh-Hora M, et al. Hair loss and defective T- and B-cell function in mice lacking ORAI1. Mol Cell Biol. 2008;28(17):5209-5222. https://doi.org/10.1128/MCB.00360-08;
[11].Vig M, DeHaven WI, Bird GS, et al. Defective mast cell effector functions in mice lacking the CRACM1 pore subunit of store-operated calcium release-activated calcium channels. Nat Immunol. 2008;9(1):89-96. https://doi.org/10.1038/ni1550.
[12].Oh-Hora M, Yamashita M, Hogan PG, et al. Dual functions for the endoplasmic reticulum calcium sensors STIM1 and STIM2 in T cell activation and tolerance. Nat Immunol. 2008;9(4):432-443. https://doi.org/10.1038/ni1574.
[13].Qu B, Al-Ansary D, Kummerow C, Hoth M, Schwarz EC. ORAI-mediated calcium influx in T cell proliferation, apoptosis and tolerance. Cell Calcium. 2011;50(3):261-269. https://doi.org/10.1016/j.ceca.2011.05.015.
[14].Matsumoto M, Fujii Y, Baba A, Hikida M, Kurosaki T, Baba Y. The calcium sensors STIM1 and STIM2 control B cell regulatory function through interleukin-10 production. Immunity. 2011;34(5):703-714. https://doi.org/10.1016/j.immuni.2011.03.016.
[15].Wu B, Ramaiah A, Garcia G Jr, Hasiakos S, Arumugaswami V, Srikanth S. ORAI1 Limits SARS-CoV-2 Infection by Regulating Tonic Type I IFN Signaling. J Immunol. 2022;208(1):74-84. https://doi.org/10.4049/jimmunol.2100742.
[16].Srikanth S, Woo JS, Wu B, et al. The Ca2+ sensor STIM1 regulates the type I interferon response by retaining the signaling adaptor STING at the endoplasmic reticulum. Nat Immunol. 2019;20(2):152-162. https://doi.org/10.1038/s41590-018-0287-8.
[17].Bryant N, Muehling LM. T-cell responses in asthma exacerbations. Ann Allergy Asthma Immunol. 2022;129(6):709-718. https://doi.org/10.1016/j.anai.2022.07.027.
[18].Vaeth M, Maus M, Klein-Hessling S, et al. Store-Operated Ca2+ Entry Controls Clonal Expansion of T Cells through Metabolic Reprogramming. Immunity. 2017;47(4):664-679.e6. https://doi.org/10.1016/j.immuni.2017.09.003.
[19].Zhu H, Ruan X, Zhao K, et al. The miR-641-STIM1 and SATB1 axes play important roles in the regulation of the Th17/Treg balance in ITP. Sci Rep. 2024;14(1):11243. Published 2024 May 16. https://doi.org/10.1038/s41598-024-61660-9.
[20].Liu CC, Miao Y, Chen RL, et al. STIM1 mediates IAV-induced inflammation of lung epithelial cells by regulating NLRP3 and inflammasome activation via targeting miR-223. Life Sci. 2021;266:118845. https://doi.org/10.1016/j.lfs.2020.118845.
[21].Usmani SM, von Einem J, Frick M, et al. Molecular basis of early epithelial response to streptococcal exotoxin: role of STIM1 and Orai1 proteins. Cell Microbiol. 2012;14(3):299-315. https://doi.org/10.1111/j.1462-5822.2011.01724.x.
[22].Fan H, Huang H, Hu L, et al. The activation of STIM1 mediates S-phase arrest and cell death in paraquat induced acute lung intoxication. Toxicol Lett. 2018;292:123-135. https://doi.org/10.1016/j.toxlet.2018.04.030.
[23].Yang W, Tian R, Zhu Y, et al. Paraquat is an agonist of STIM1 and increases intracellular calcium levels. Commun Biol. 2022;5(1):1151. Published 2022 Oct 30. https://doi.org/10.1038/s42003-022-04130-0.
[24].Song X, Liu Y, Dong L, Wang Y. Stromal-Interacting Molecule 1 (Stim1)/Orai1 Modulates Endothelial Permeability in Ventilator-Induced Lung Injury. Med Sci Monit. 2018;24:9413-9423. Published 2018 Dec 27. https://doi.org/10.12659/MSM.911268.
[25].Yuan XH, Li YM, Shen YY, Yang J, Jin Y. Clinical and Th1/Th2 immune response features of hospitalized children with human rhinovirus infection. J Med Virol. 2020;92(1):26-33. https://doi.org/10.1002/jmv.25587.
[26].Thomas R, Qiao S, Yang X. Th17/Treg Imbalance: Implications in Lung Inflammatory Diseases. Int J Mol Sci. 2023;24(5):4865. Published 2023 Mar 2. https://doi.org/10.3390/ijms24054865.
Peer-review Terminology
Identity transparency: Single anonymized
Reviewer interacts with: Editor
Details
This is an open access article under the terms of the Creative Commons Attribution License(http://creativecommons.org/licenses/by/4.0/), which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Publication History
Received 2025-04-02
Accepted 2025-05-09
Published 2025-05-15


