Abstract
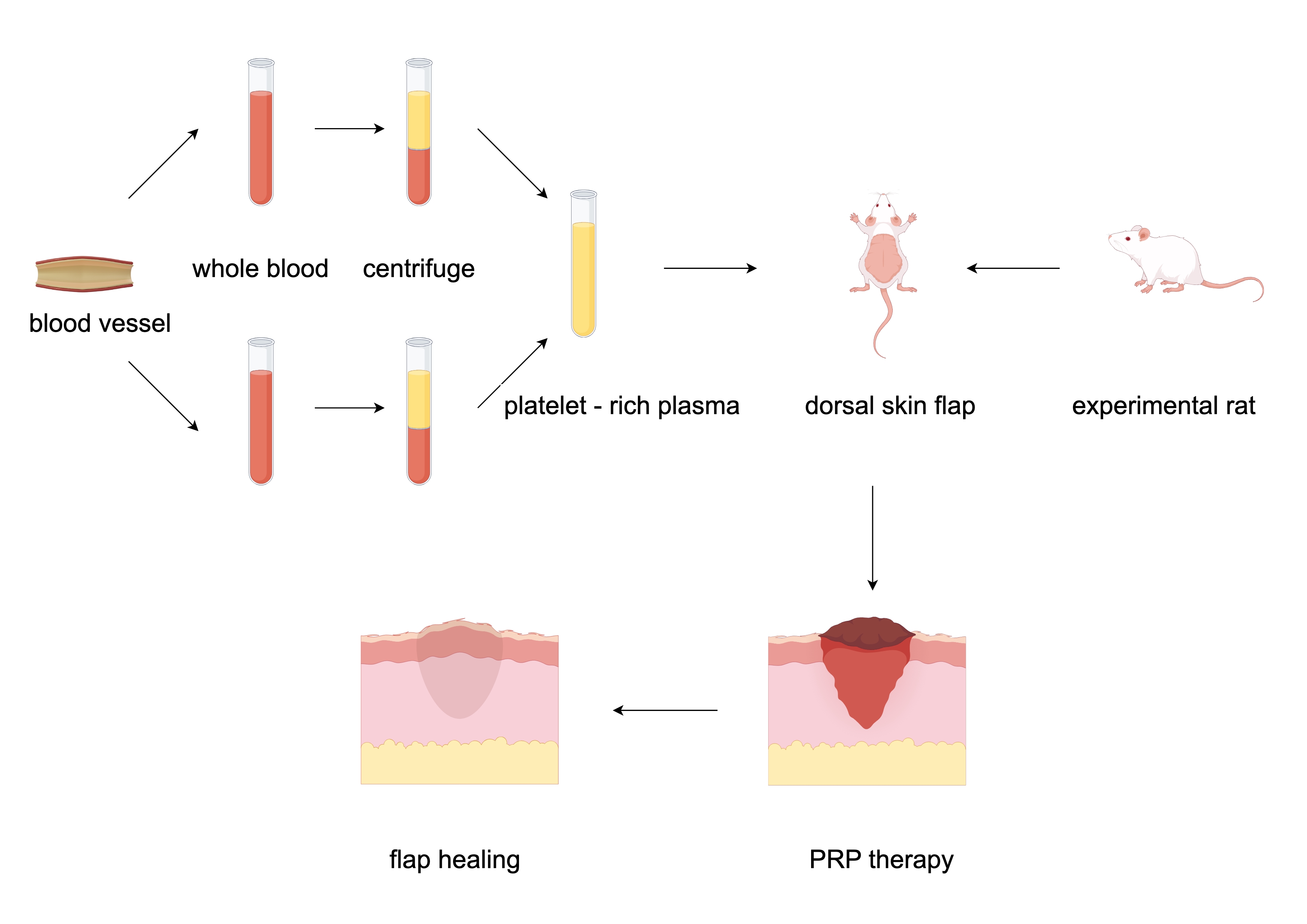
Background: Investigating the Effects and Mechanisms of Platelet-Rich Gel (PRG) Combined with Platelet-Rich Plasma (PRP) on the Transplantation of Dorsal Skin Flaps in SD Rats.
Method: A rat dorsal pedicle skin flap model was established, and 84 SD rats were randomly divided into 14 groups: the control group, the PRG topical treatment group, and the PRG+PRP combined treatment group. The control group received no intervention. The PRG group was treated with PRG at doses of 50 μl/cm² and 100 μl/cm² applied to the base. The combined group received PRG application followed by PRP injection at the base, with treatments including 50 μl/cm² PRG + 50 μl/cm² PRP, 100 μl/cm² PRG + 50 μl/cm² PRP, 50 μl/cm² PRG + 100 μl/cm² PRP, and 100 μl/cm² PRG + 100 μl/cm² PRP. On postoperative days 3 and 5, the necrosis ratio of the skin flap, the defect length measured by ultrasound contrast, and the degree of edema were recorded. On postoperative day 3, skin flap tissues from each group were collected for VEGF and CD34 immunohistochemical staining. The expression levels were scored, and the number of microvessels was counted based on CD34 staining.
Result: On postoperative days 3 and 5, the 50 μl/cm² PRG + 50 μl/cm² PRP group exhibited the lowest skin flap necrosis ratio and the shortest defect length measured by ultrasound contrast, both of which were significantly lower than those in the control group (P < 0.05). On postoperative day 3, the expression levels of VEGF and CD34, as well as the number of microvessels in this group, were significantly higher than those in the control group (P < 0.05).
Conclusion: The 50 μl/cm² PRG + 50 μl/cm² PRP group can achieve the optimal improvement in the post-transplantation condition of skin flaps by enhancing the expression of VEGF and CD34 in the flap tissue and promoting angiogenesis.
Keywords: Platelet-Rich Plasma; Platelet-Rich Gel; Skin Flap; Ultrasound Contrast;
Introduction
Flap transplantation is a crucial technique in reconstructive surgery for wound repair[1], due to the disruption of the original blood supply pattern in the flap area during the transplantation process, necrosis in the distal region of the flap has become one of the most common postoperative complications[2]. Platelet-rich plasma (PRP) and platelet-rich gel (PRG) are both concentrated platelet products derived from whole blood, known for their abilities to reduce inflammatory responses and ischemia-reperfusion injury, as well as promote tissue repair[3,4], which are widely used in fields such as orthopedics, oral and wound repair surgery[5].
Both PRP and PRG share the same active components; however, compared to PRP, PRG can induce degranulation more rapidly, releasing a large amount of growth factors to promote blood supply reconstruction in the flap area[6]. However, the mechanical instability of PRG and its difficulty in adhering to the wound surface have hindered the therapeutic efficacy of PRP.
To address this, the research team combined PRP with PRG at varying doses in a rat dorsal pedicle skin flap model to observe their effects on flap transplantation outcomes. The study aimed to determine the optimal combined dosage of PRG and PRP and elucidate their underlying mechanisms, thereby providing a theoretical foundation for the clinical application of PRG and PRP in combination.
Materials and Methods
Experimental Animals
Healthy SPF-grade SD female rats, 154 in total, aged 7-8 weeks, weighing 210-240g. Among them, 84 rats were used for experimental modeling, and 70 rats were used for blood collection to prepare PRP. The rats were purchased from the Experimental Animal Center of Anhui Medical University (License:SYXK(Wan)2017-006) and housed under conditions of room temperature 20°C–28°C and humidity 45%–75%, with 3 rats per cage and free access to food and water. The experiment was approved by the Animal Ethics Committee of Anhui Medical University (Approval No.: LLSC20241735).
Main Reagents and Instruments
80°C Ultra-Low Temperature Freezer, Model DW-HL5, purchased from Zhongke Meiling Cryogenic Technology.Low-Speed Benchtop Centrifuge, Model L3-5K, purchased from Hunan Kecheng Instrument Equipment Co., Ltd.Ultrasound Diagnostic Instrument, Model ACUSON Sequoia, purchased from Siemens Healthcare GmbH.Enhanced Small Animal Anesthesia Machine, Model R540, purchased from Shenzhen Reward Life Technology Co., Ltd.Automatic Blood Cell Analyzer, Model BC-5390 CRP, purchased from Mindray Medical International Co., Ltd.Thermal Imaging Camera, Model E3PLUS, purchased from Shenzhen Arrow Electronics Co., Ltd.High-Resolution Biological Microscope, Model DP74, purchased from Olympus (China) Co., Ltd.VEGF Antibody Reagent and CD34 Antibody Reagent, both purchased from Fuzhou Maixin Biotech Co., Ltd.Thrombin Lyophilized Powder, purchased from Changchun Leiyunshang Pharmaceutical Co., Ltd.Calcium Gluconate Injection, purchased from Sichuan Medcome Pharmaceutical Co., Ltd.Sulfur Hexafluoride Microbubble Injection, purchased from Bracco Suisse SA.
Experimental Grouping
The experiment was divided into two time points: post-operative day 3 and post-operative day 5. At each time point, the groups included a control group, a PRG group, and a PRG combined with PRP group. The PRG group was further subdivided by dose into 0.5G and 1G groups, while the PRG combined with PRP group was divided into 0.5P0.5G, 0.5P1G, 1P0.5G, and 1P1G groups (0.5G and 1G represent PRG doses of 50μl/cm² and 100μl/cm², respectively; 0.5P and 1P represent PRP doses of 50μl/cm² and 100μl/cm², respectively). A total of 7 groups were established, with 6 rats in each group.
PRG Preparation
Two rats were randomly selected from 70 blood-donor rats to extract whole blood for the preparation of PRP and PRG. Throughout the process, ensure aseptic techniques are followed to prevent contamination of the PRG. The specific operational steps are as follows:
Whole Blood Extraction.Rats were anesthetized with 3.5% isoflurane inhalation and fixed in a supine position.The abdominal area was disinfected, and the skin was incised along the linea alba.The intestines were gently moved aside to expose the inferior vena cava.A 0.55 mm diameter blood collection needle was used to draw 5 ml of whole blood, which was then transferred to an anticoagulant tube.The rats were euthanized following blood collection.PRP Extraction: PRP was extracted using the Landesberg modified method[7] .
Place the freshly collected whole rat blood into a centrifuge and centrifuge at 200 g for 20 minutes.Use a 1 mL syringe to carefully extract the supernatant and leukocyte layer, avoiding the red blood cells as much as possible. Transfer the extracted supernatant and leukocyte layer into a new dry tube and centrifuge again at 200 g for 10 minutes. After the second centrifugation, discard the upper 3/5 of the plasma. The remaining plasma is mixed thoroughly to obtain PRP. Take 0.2 mL of the PRP in a sterile EP tube and use the BC-5390 CRP blood cell analyzer to count the platelet concentration in the sample.
PRG Preparation. Prepare the PRP activator by mixing thrombin freeze-dried powder with 10% calcium gluconate injection at a ratio of 1000 U:1 mL. Mix the PRP with the activator at a volume ratio of 10:1 to obtain PRG.
Animal Model Creation
Based on the anatomical characteristics of the dorsal blood vessels in rats[8] , the research team utilized the unilateral thoracodorsal artery as the blood supply artery to design a dorsal flap model measuring 1 cm in length and 6.5 cm in width. Considering the variations in dorsal vascular distribution among different rats, the experiment selected rats with body weights within the range of 220–240 g, excluding individuals with missing intercostal arteries or those exhibiting two intercostal arteries.
Flap Design
Landmark Identification:Identify point A as the intersection of the posterior midline and the line connecting the inferior angles of the scapulae on both sides. Mark point B on the posterior midline, 1 cm below point A, and point C, 7.5 cm below point A. Lateral Points:From points B and C, draw lines parallel to the line connecting the inferior angles of the scapulae, extending 2 cm laterally to mark points D and E, respectively. Midpoints:Locate the midpoints of BD and CE, labeled as F and G, respectively. Flap Area Formation: Connect points D, E, G, and F to form a rectangular flap area measuring 1 cm × 6.5 cm.
Figure 1 Flap design and fabrication schematic diagram.
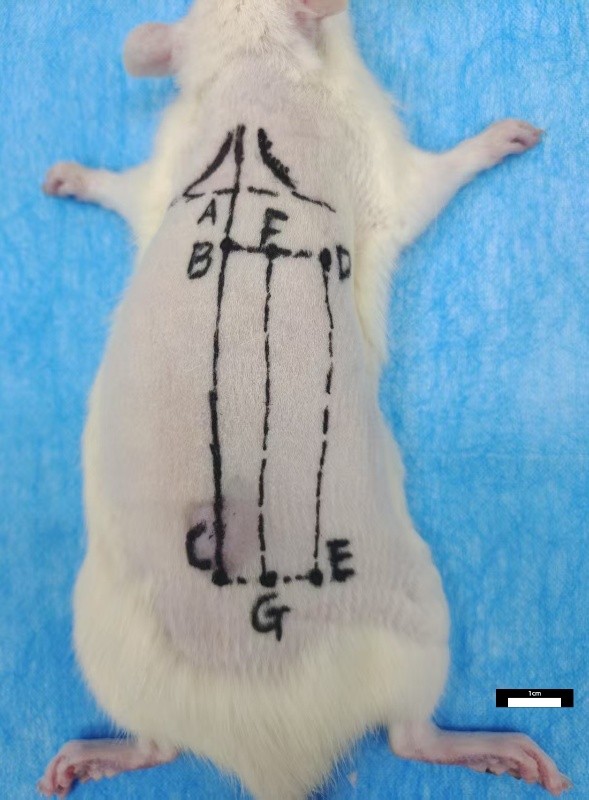
Flap Preparation
After inducing anesthesia with 3.5% isoflurane for 3 minutes, adjust the isoflurane concentration to the maintenance level of 2%. Fix the rat in a prone position, and perform shaving, disinfection, and draping on the rat’s back. Incise the skin and superficial fascia layer along the marked line, and use tissue scissors to separate the superficial fascia layer from the deep fascia layer. Preserve only the thoracodorsal artery perforator as the blood supply artery for the flap. Both the experimental group and the blank control group rats were designed with this flap model.
Flap Treatment
During the procedure, the corresponding unit volume of PRG is evenly applied between the flap and the base for drug administration; PRP is administered through multiple injections at the base. The control group receives no treatment, and the flap is simply sutured in place with full-thickness stitches.
Flap Necrosis Ratio
On the 3rd and 5th days post-operation, the necrotic conditions at the distal ends of the flaps in the 3-day and 5-day groups were recorded using a camera. Necrosis was defined as obvious whitening, edema, black scabbing, hardening of texture, loss of elasticity, and absence of bleeding upon needle puncture. The captured images were analyzed using Image Pro Plus 6.0 software to calculate the flap necrosis ratio (necrosis ratio = necrotic area / total flap area). Statistical analysis was performed on the flap necrosis ratios of each group.
External Jugular Vein Cannulation and Contrast-Enhanced Ultrasound
On the 3rd and 5th days post-operation, external jugular vein cannulation and contrast-enhanced ultrasound were performed on the 3-day and 5-day groups of rats, respectively. After anesthesia, the rats were fixed in a supine position. The skin on one side of the neck was shaved, disinfected, and draped, and the external jugular vein was isolated. A 24G indwelling needle was used for cannulation and fixation. Under ultrasound guidance at a frequency of 10 MHz, 0.5 mL of sulfur hexafluoride microbubble contrast agent was injected through the indwelling needle to perform contrast-enhanced ultrasound. The length of the perfusion defect in the dorsal tissue flap and the skin thickness at the distal 1 cm of the flap were recorded.
Platelet Count and Tissue Flap Sampling
On the 3rd and 5th days post-operation, venous whole blood and tissue samples from the distal 0.5 cm to 1.5 cm of the flap were collected from the 3-day and 5-day groups of rats, respectively. The number of platelets in the whole blood samples was counted using a hematology analyzer, and the concentration factor of platelets in PRP and PRG was calculated (concentration factor = platelet count in PRP or PRG / platelet count in whole blood). The tissue flap samples were bisected along the horizontal midline. The distal 0.5 cm to 1 cm portion of the flap was fixed in 10% formalin and stored in a cool, dry place, while the distal 1 cm to 1.5 cm portion was frozen and stored at -80°C.
HE Staining
On the 3rd and 5th days post-operation, the skin tissue layers of the rats in each group were embedded in paraffin, sectioned into slides, and subjected to HE staining. Under a microscope, the necrosis and shedding of epithelial cells, the degree of inflammatory cell infiltration, and the aggregation of inflammatory cells in the rat skin tissue layers were observed.
Immunohistochemistry and Vascular Counting
Immunohistochemistry
Paraffin-embedded tissue sections from the rat flaps on the 3rd day post-operation were prepared and subjected to immunohistochemical staining to observe the expression of vascular endothelial growth factor (VEGF) and CD34 in the flaps of each group. A semi-quantitative method was used to score the staining results. For each satisfactory stained section, six random 400x magnification fields were selected for scoring, and the average of the six scores was taken as the final score. Positive staining was defined as the presence of brownish-yellow or brown granules localized in the cytoplasm or cell membrane, and the percentage of positive cells relative to the total skin tissue cells was counted. The specific scoring criteria were as follows: Staining intensity: No staining = 0 points; light yellow = 1 point; light brown = 2 points; dark brown = 3 points. Percentage of positive cells: 0% = 0 points; 1%–24% = 1 point; 25%–49% = 2 points; 50%–74% = 3 points; 75%–100% = 4 points. The final score was calculated as the product of the staining intensity score and the positive cell percentage score. Statistical analysis was performed on the final scores of each group.
Microvessel Counting
Under a 400x magnification field of CD34 immunohistochemical staining, six discontinuous regions were randomly selected to count the number of microvessels. The average value was calculated as the vascular density of the sample. Statistical analysis was performed on the number of vessels in each group of rats.
Statistical Methods
Statistical analysis was performed using SPSS 24.0 software. Measurement data conforming to a normal distribution were expressed as mean ± standard deviation(±s). Intergroup comparisons were conducted using one-way ANOVA. For data with homogeneity of variance, pairwise comparisons were performed using the Bonferroni method, while for data with heterogeneity of variance, the Games-Howell method was used. A P-value of <0.05 was considered statistically significant.
Results
Group Balance Test
Comparison of Rat Body Weight Among Groups
The experimental animals used in this study were healthy SPF-grade female SD rats, aged 7-8 weeks, with a weight range of 220~240 g. There was no statistically significant difference in the mean body weight of rats among the groups(F =1.376,P =0.193).
Comparison of Platelet Concentration Factors Among Groups
The platelet counts of the prepared platelet-rich products and the whole blood of the model rats were measured using an automated hematology analyzer. There was no statistically significant difference in the mean platelet concentration factors (platelet count in platelet-rich products / platelet count in whole blood) among the groups(F =1.233,P =0.286).
Flap Distal Necrosis Ratio
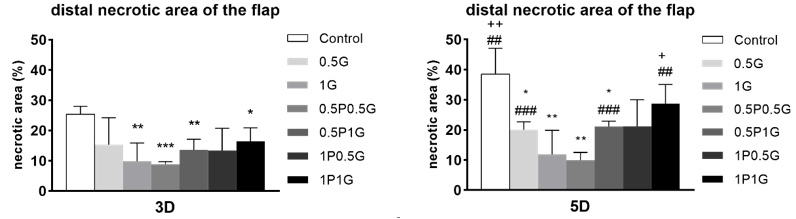
On the 3rd day post-operation, distal necrosis changes were observed in the flaps of rats in all groups, and the necrotic area gradually increased over time. By the 5th day post-operation, the extent of distal necrosis in the skin tissue layers of rats in each group tended to stabilize. Using Image Pro Plus 6.0, the necrosis ratio of the flaps post-operation was calculated. On the 3rd day, the necrosis ratio in the control group and the PRG group was significantly higher than that in the 1G group, and the difference was statistically significant(P <0.05); Compared to the control group and the combined treatment group, the necrosis ratio was significantly higher in the 0.5P0.5G, 0.5P1G, and 1P1G groups, and the differences were statistically significant(P <0.05); There were no statistically significant differences among the experimental groups(P >0.05). On the 5th day post-operation, the necrosis ratio in the control group and the PRG group was significantly higher than that in the 0.5G and 1G groups, and the difference was statistically significant(P <0.05); Compared to the control group and the combined treatment group, the necrosis ratio was significantly higher in the 0.5P0.5G and 0.5P1G groups, and the differences were statistically significant(P <0.05); Among the experimental groups, the necrosis proportion in the 1G group was significantly lower than that in the 1P1G group, and the necrosis proportion in the 0.5P0.5G group was significantly lower than that in the 0.5G, 0.5P1G, and 1P1G groups, with the differences being statistically significant (P <0.05). There were no statistically significant differences among the other groups(P >0.05).(Figure 2)
Figure 2 Necrotic Area Analysis Diagram (X̅±s,%)
Skin Tissue Layer Ultrasound Contrast Defects
According to the ultrasound contrast results, varying degrees of filling defects were observed in the dorsal skin flaps of rats on the third day post-surgery, with a rising trend over time. On the third day post-surgery, the defect length at the flap end in the control group and the PRG group was significantly greater than that in the 1G group, with a statistically significant difference (P < 0.05). Compared to the combined treatment group, the defect length at the flap end in the control group was significantly greater than that in the 0.5P0.5G, 0.5P1G, and 1P0.5G groups, with a statistically significant difference (P < 0.05). Among the experimental groups, the defect length at the flap end in the 1G and 0.5P0.5G groups was significantly smaller than that in the 0.5G group, with a statistically significant difference (P < 0.05). No statistically significant differences were observed among the remaining groups.
On the fifth day post-surgery, the defect length at the flap end further increased in all groups. The defect length in the control group was significantly greater than that in the 0.5P0.5G group, with a statistically significant difference (P < 0.05). Among the experimental groups, the defect length in the 0.5G group was significantly greater than that in the 1G, 0.5P0.5G, 0.5P1G, and 1P0.5G groups, with a statistically significant difference (P< 0.05). No statistically significant differences were observed among the remaining groups.(Table 1)
Table 1 Statistical table of the defect length in skin flaps (X̅±s,mm)
|
Group |
Number |
The 3rd day post-operation |
The 5th day post-operation |
|
Contral |
6 |
4.50±1.03 |
7.75±3.26 |
|
0.5G |
6 |
3.08±0.08 |
4.55±0.70 |
|
1G |
6 |
1.70±0.72①③ |
2.23±0.95③ |
|
0.5P0.5G |
6 |
1.43±0.90①③ |
1.73±1.26②③ |
|
0.5P1G |
6 |
2.05±0.79② |
2.55±1.01③ |
|
1P0.5G |
6 |
1.92±0.97② |
2.40±0.99③ |
|
1P1G |
6 |
2.87±0.66 |
3.47±1.39 |
|
F |
|
13.548 |
15.392 |
|
P |
|
<0.001 |
<0.001 |
Note: The intergroup comparison of this dataset was performed using the Games-Howell method; ① indicates P <0.01 compared to the control group; ② indicates P <0.05 compared to the control group; ③ indicates P <0.05 compared to the 0.5G group.
Skin Tissue Layer Edema Degree
The edema condition of the skin flaps in rats post-surgery can be reflected by the thickness difference between the flap end tissue and the surrounding normal skin. According to the 10MHz ultrasound measurement results, except for the 1P1G group, the edema at the flap end in all groups decreased on the fifth day post-surgery compared to the third day.
On the third day post-surgery, the edema degree at the flap end in the control group and the PRG group was significantly greater than that in the 1G group, with a statistically significant difference (P < 0.05). Compared to the combined treatment group, the edema degree in the control group was significantly greater than that in the 0.5P0.5G and 1P0.5G groups, with a statistically significant difference (P < 0.05). Among the experimental groups, the edema degree in the 0.5P0.5G group was significantly smaller than that in the 1P1G group, with a statistically significant difference (P < 0.05). No statistically significant differences were observed among the remaining groups(P > 0.05).
On the fifth day post-surgery, the edema degree at the flap end in the 1P1G group was significantly greater than that in the control group and all other experimental groups, with a statistically significant difference (P < 0.05). No statistically significant differences were observed among the remaining groups (P > 0.05).(Table 2)
Table 2 Statistical table of the edema degree at the flap (X̅±s,mm)
|
Group |
Number |
The 3rd day post-operation |
The 5th day post-operation |
|
Contral |
6 |
0.40±0.06 |
0.30±0.06⑤ |
|
0.5G |
6 |
0.25±0.10 |
0.23±0.10③ |
|
1G |
6 |
0.22±0.08② |
0.20±0.06③ |
|
0.5P0.5G |
6 |
0.17±0.08①⑤ |
0.15±0.10③ |
|
0.5P1G |
6 |
0.28±0.08 |
0.27±0.10④ |
|
1P0.5G |
6 |
0.23±0.08② |
0.22±0.08③ |
|
1P1G |
6 |
0.33±0.10 |
0.47±0.08 |
|
F |
|
5.055 |
8.323 |
|
P |
|
<0.001 |
<0.001 |
Note: The intergroup comparison of this dataset was performed using the Bonferroni method; ① indicates P < 0.001 compared to the control group; ② indicates P < 0.05 compared to the control group; ③ indicates P < 0.001 compared to the 1P1G group; ④ indicates P < 0.01 compared to the 1P1G group; ⑤ indicates P < 0.05 compared to the 1P1G group.
HE Staining Results
Based on the HE staining results of rat skin flaps post-surgery, inflammatory cell infiltration and epidermal detachment were observed in all groups, with a worsening trend over time. By the fifth day post-surgery, the flaps exhibited chronic inflammation. On the third day post-surgery, the number of inflammatory cells in the 1G, 0.5P0.5G, 0.5P1G, and 1P0.5G groups decreased compared to the control group, and the degree of epidermal detachment was reduced. On the fifth day post-surgery, the 0.5P0.5G group maintained a relatively lower level of inflammatory cells compared to the other groups, and epithelial cells were better preserved.
Immunohistochemistry Results
The staining results of each group are shown in Figure 3. On the third day post-operation, VEGF and CD34 immunohistochemical staining was performed on the skin flap tissues of the rats in each group, and the staining results were scored. The results showed that compared with the PRG group, the VEGF expression in the control group was significantly lower than that in the 0.5G group, and the CD34 expression was significantly lower than that in the 1G group, with statistically significant differences (P < 0.05). Compared with the combination therapy group, the VEGF and CD34 expressions in the control group were significantly lower than those in the 0.5P0.5G and 0.5P1G groups, with statistically significant differences (P < 0.05). Among the experimental groups, the VEGF expression in the 1P0.5G group was significantly higher than that in the 1P1G group, and the CD34 expression in the 0.5P0.5G group was significantly higher than that in the 1P0.5G and 1P1G groups, with statistically significant differences (P < 0.05). There were no statistically significant differences among the other groups.(Table 3)
Figure 3 The schematic diagram of immunohistochemical staining results

Table 3 The statistical table of VEGF and CD34 immunohistochemical scores (X̅±s)
|
Group |
Number |
VEGF |
CD34 |
||||
|
|
|
Intensity |
Percentage |
Total |
Intensity |
Percentage |
Total |
|
Contral |
6 |
0.83±0.75 |
0.83±0.75 |
1.00±0.89 |
0.83±0.75 |
0.83±0.75 |
1.00±0.89 |
|
0.5G |
6 |
2.00±0.63 |
1.83±0.75 |
3.33±0.82② |
1.83±1.17 |
1.17±0.75 |
2.33±1.37 |
|
1G |
6 |
2.17±0.75 |
2.33±0.82 |
5.00±2.53 |
2.33±0.82 |
1.83±0.75 |
3.83±1.17② |
|
0.5P0.5G |
6 |
2.33±0.82 |
2.33±0.82 |
5.17±2.32② |
2.33±0.82 |
2.17±0.75 |
4.83±2.32① |
|
0.5P1G |
6 |
2.17±0.75 |
2.00±0.63 |
4.00±1.10①③ |
2.33±0.82 |
1.83±0.75 |
4.00±1.67② |
|
1P0.5G |
6 |
1.33±1.03 |
1.17±0.75 |
1.67±1.03 |
1.50±1.38 |
0.83±0.75 |
1.67±1.37④ |
|
1P1G |
6 |
2.00±0.63 |
1.67±0.82 |
3.33±1.75 |
1.50±1.05 |
1.00±0.63 |
1.67±1.03④ |
|
F |
|
- |
- |
7.161 |
|
|
5.855 |
|
P |
|
- |
- |
<0.001 |
|
|
<0.001 |
Note: The VEGF intergroup comparison was performed using the Games-Howell method; the CD34 intergroup comparison was performed using the Bonferroni method; ① indicates P < 0.01 compared to the control group; ② indicates P < 0.05 compared to the control group; ③ indicates P < 0.05 compared to the 1P0.5G group; ④ indicates P < 0.05 compared to the 0.5P0.5G group.
Microvessel Count
Under the CD34 staining background, microvessel counts were performed on the rat skin flaps. It was observed that microvessels had formed in the skin flap tissues on the third day post-operation. On the third day post-operation, compared to the control group and the monotherapy group, the number of microvessels in the skin flap tissues was significantly lower in the 1G group, with a statistically significant difference (P < 0.05). Compared to the control group and the combination therapy group, the number of microvessels in the skin flap tissues was significantly lower in the 0.5P0.5G, 0.5P1G, and 1P0.5G groups, with statistically significant differences (P < 0.05). Among the experimental groups, the number of capillaries in the skin flap tissues in the 1P0.5G group was significantly lower than that in the 1G and 0.5P0.5G groups, with statistically significant differences (P < 0.05). (Table 4)
Table 4 Statistics of Microvessel Count in Flap Tissue (X̅±s)
|
Group |
Number |
Microvessel Count |
|
contral |
6 |
2.67±0.82 |
|
0.5G |
6 |
4.00±0.63 |
|
1G |
6 |
6.83±0.75①③ |
|
0.5P0.5G |
6 |
8.00±0.89①② |
|
0.5P1G |
6 |
6.17±0.75① |
|
1P0.5G |
6 |
5.00±0.89① |
|
1P1G |
6 |
4.00±0.63 |
|
F |
|
34.616 |
|
P |
|
<0.001 |
Note: Intergroup comparison of microvessel count was performed using the Bonferroni method; ① indicates P < 0.001 compared to the control group; ② indicates P < 0.001 compared to the 1P0.5G group; ③ indicates P < 0.01 compared to the 1P0.5G group.
Discussion
Flap necrosis involves physiological mechanisms primarily including tissue circulation disorders and ischemia-reperfusion injury[9,10] . In the early postoperative period of flap surgery, the original blood supply system in the flap area is disrupted, leading to insufficient blood perfusion in the distal part of the flap and varying degrees of venous reflux obstruction. This results in inadequate oxygen and nutrient supply to the flap tissue and the inability to timely remove metabolic waste products, triggering the generation of oxygen free radicals, dysfunction of the sodium-potassium pump, vascular spasm, and intravascular thrombosis[11,12] . As the postoperative period progresses, the establishment of collateral circulation and vascular regeneration increases tissue blood perfusion. Ischemia-reperfusion injury further promotes the generation of oxygen free radicals, triggering the synthesis of pro-inflammatory mediators. Activated pro-inflammatory mediators enhance the effects of various cytokines and chemokines, promoting leukocyte migration and inducing the release of tissue-damaging enzymes, ultimately leading to widespread cellular apoptosis and necrosis[13] .
PRP (Platelet-Rich Plasma) is a concentrated platelet product extracted from whole blood through gradient centrifugation. PRG (Platelet-Rich Gel) is a gel-like substance made by mixing PRP with an activator in a certain ratio. Both are platelet-rich products containing a large number of growth factors, such as PDGF, VEGF, TGF-β, insulin-like growth factor-1, and epidermal growth factor. They can help flap survival by accelerating and promoting flap angiogenesis[14–16] . Due to the limited blood volume of rats, this experiment utilized allogeneic rat blood to prepare PRP and PRG. The platelet count in the platelet-rich products obtained in this study was (2603.58 ± 363.74) × 10^9/L, which is (3.51 ± 0.44) times that of whole blood platelets. No significant immune rejection reactions were observed during the application of allogeneic platelet-rich products in rat flaps, which aligns with the findings reported by Akbarzadeh S et al[17] .
In this study, the individual treatment group applied different unit volumes of PRG to the base of the flap, while the combined treatment group combined PRG application with injections of different unit volumes of PRP to observe the effects on rat dorsal flap transplantation. The results showed that the proportion of flap necrosis in the experimental groups was improved compared to the control group at all postoperative time points. When PRG was used alone, the 100μl/cm² group showed more significant effects in reducing flap necrosis compared to the 50μl/cm² group. When PRP and PRG were used in combination, both the 50μl/cm² PRP + 50μl/cm² PRG group and the 50μl/cm² PRP + 100μl/cm² PRG group exhibited significantly less flap necrosis than the control group on the third and fifth postoperative days. Among all experimental groups, the 50μl/cm² PRP + 50μl/cm² PRG group showed the most significant improvement in flap necrosis. Further increasing the dosage of PRP or PRG did not contribute to additional improvement in flap necrosis, which may be related to the characteristics of PRP and PRG and their different administration methods. PRP is in liquid form and is directly produced by secondary centrifugation of whole blood, while PRG is made by mixing PRP with a certain proportion of activator. In the experiment, PRP was administered via multiple injections at the base, whereas PRG was applied by spreading between the flap and the base. Compared to PRP, PRG can more rapidly release a large amount of growth factors through degranulation, aiding in the faster reconstruction of blood circulation in the postoperative flap area. However, PRG is prone to liquefaction and may be lost from the wound, preventing it from functioning stably. The multiple injections of PRP effectively compensate for this limitation.
Contrast Enhanced Ultrasonography (CEUS) is a clinical examination technique that visualizes the blood perfusion of target organs by intravenously injecting sulfur hexafluoride microbubbles[18] .It can not only effectively reflect the blood perfusion of the flap, but also measure the thickness of the flap and normal skin in contrast mode to assess the degree of postoperative edema in the flap. In this study, we observed that the length of flap contrast defects in the experimental group rats at various postoperative time points showed a decreasing trend compared to the control group. When PRG was used alone, the 100μl/cm² group had a more significant effect in reducing flap necrosis compared to the 50μl/cm² group. When PRP and PRG were used in combination, the 50μl/cm² PRP + 50μl/cm² PRG group, the 50μl/cm² PRP + 100μl/cm² PRG group, and the 100μl/cm² PRP + 50μl/cm² PRG group showed significantly smaller flap contrast defects than the control group on the third postoperative day. By the fifth postoperative day, only the 50μl/cm² PRP + 50μl/cm² PRG group still had a significant difference in contrast defect length compared to the control group among all experimental groups. Additionally, on the third postoperative day, the 100μl/cm² PRG alone treatment group, the 50μl/cm² PRP + 50μl/cm² PRG combination treatment group, and the 100μl/cm² PRP + 50μl/cm² PRG group showed significant improvement in flap end edema compared to the control group, with the 50μl/cm² PRP + 50μl/cm² PRG group showing the most significant improvement in edema. By the fifth postoperative day, only the 100μl/cm² PRP + 100μl/cm² PRG group showed further increase in flap edema, while the edema in the other groups had reduced and showed no significant differences. This indicates that the 50μl/cm² PRP + 50μl/cm² PRG group not only performed more stably in improving flap contrast defects but also maximally reduced postoperative flap edema. Moreover, the increased edema in the 100μl/cm² PRP + 100μl/cm² PRG group suggests that excessive use of platelet-rich products may exacerbate local tissue edema and effusion, adversely affecting local blood return and tissue metabolic activity. Compared to visual observation of flap conditions, CEUS can more accurately reflect postoperative blood supply in the flap, allowing for a reasonable prediction of the occurrence and progression of flap necrosis. Therefore, in the early stages after clinical flap transplantation, even if no obvious necrosis is observed visually, when CEUS detects filling defects in the flap, intervention with platelet-rich products in the surrounding tissue or base of the flap may improve or even reverse the process of flap necrosis. This hypothesis requires further validation.
After flap transplantation, the flap tissue often experiences phenomena such as damage to internal blood vessels, occlusion of numerous capillaries, and reduced expression of active substances like growth factors. The pathological immunohistochemistry results from this study showed that on the third postoperative day, varying numbers of newly formed capillaries could be observed in all groups under CD34 staining, with the experimental groups exhibiting an upward trend in capillary count compared to the control group. Specifically, the 100μl/cm² PRG alone treatment group, the 50μl/cm² PRP + 50μl/cm² PRG group, the 100μl/cm² PRP + 50μl/cm² PRG group, and the 50μl/cm² PRP + 100μl/cm² PRG group showed significantly higher numbers of newly formed capillaries compared to the control group. Among the experimental groups, the 100μl/cm² PRG alone treatment group and the 50μl/cm² PRP + 50μl/cm² PRG group had notably higher capillary counts than the 50μl/cm² PRP + 100μl/cm² PRG group, with the latter showing more significant differences. These results align with the CD34 immunohistochemistry staining results of the flap tissue and are largely consistent with the VEGF immunohistochemistry staining results. This indicates that PRP and PRG can enhance the expression of VEGF and CD34 in flap tissue to promote angiogenesis, with the 50μl/cm² PRP + 50μl/cm² PRG group showing the most pronounced effect on angiogenesis.
This study demonstrates that PRP and PRG can promote flap survival by upregulating the expression of VEGF and CD34 in flap tissue, thereby enhancing angiogenesis. When different volumes of allogeneic PRG were applied alone to rat dorsal flap transplantation surgery, the 100μl/cm² group showed more significant effects in promoting angiogenesis, reducing postoperative flap edema, and decreasing the proportion of flap necrosis and contrast defect length compared to the 50μl/cm² group. In the groups treated with PRP combined with PRG, the group with both doses at 50μl/cm² showed the best effect in promoting flap survival, and increasing the doses did not further enhance flap survival. When both doses reached 100μl/cm², the expression of VEGF and CD34 in the flap decreased postoperatively, the number of newly formed capillaries reduced, and the degree of edema worsened, leading to increased contrast defect length and necrosis proportion in the flap. Compared to the 100μl/cm² PRG alone treatment, the 50μl/cm² PRP injection combined with 50μl/cm² PRG application showed no significant difference in promoting angiogenesis, reducing postoperative flap edema, and decreasing the proportion of flap necrosis and contrast defect length, but the statistical difference in treatment effect was more pronounced when compared to the control group.
In summary, the 50μl/cm² PRP injection combined with 50μl/cm² PRG application can maximally promote angiogenesis, reduce postoperative flap edema, and decrease the proportion of flap necrosis and contrast defect length. This study provides a theoretical basis for the clinical use of PRP combined with PRG to improve the success rate of flap transplantation and advances the establishment of standardized guidelines for the use of PRP and PRG.
Acknowledgements
Not applicable.
Author contributions
Conceptualization: Fang Linsen; Chen Qingyuan
Methodology: Fang Linsen; Chen Qingyuan
Software: Chen Qingyuan
Validation: Chen Qingyuan
Formal analysis: Chen Qingyuan
Investigation: Chen Qingyuan;Han Feng
Resources: Fang Linsen
Data curation: Chen Qingyuan;Han Feng
Writing – original draft: Chen Qingyuan
Writing – review & editing: Chen Qingyuan
Visualization: Chen Qingyuan
Supervision: Fang Linsen
Project administration: Fang Linsen
Funding acquisition: Fang Linsen
Ethics approval and consent to participate
Not applicable.
Funding information
Clinical Medicine Peak Discipline Construction Project of Anhui Medical University (Grant No. 9301001815)
Competing Interests
The authors declare that they have no existing or potential commercial or financial relationships that could create a conflict of interest at the time of conducting this study.
Data Availability
All data needed to evaluate the conclusions in the paper are present in the paper or the Supplementary Materials. Additional data related to this paper may be requested from the authors.
References
[1]KOUNIAVSKI E, EGOZI D, WOLF Y. (2022). Techniques and Innovations in Flap Engineering: A Review. Plast Reconstr Surg Glob Open, 10(9): e4523. https://doi.org/10.1097/gox.0000000000004523
[2]MYERS M B, CHERRY G. (1968). Causes of necrosis in pedicle flaps. Springer. Spec. Surg. S., 42(1): 43-50. https://pubmed.ncbi.nlm.nih.gov/4875691
[3]COLLINS T, ALEXANDER D, BARKATALI B. (2021). Platelet-rich plasma: a narrative review. EFORT Open Rev, 6(4): 225-235. https://doi.org/10.1302/2058-5241.6.200017
[4]XIE K, HUANG M, ZHENG Y, et al. (2022). Effect of Antilogous Platelet-Rich Plasma on the Revascularization of Rabbit Prefabricated Flap. Med Sci Monit, 28: e937718. https://doi.org/10.12659/MSM.937718
[5]YUAN T, GUO S C, HAN P, et al. (2012). Applications of leukocyte- and platelet-rich plasma (L-PRP) in trauma surgery. Current Pharm. Biotechnol,13(7):1173-1184. https://doi.org/10.2174/138920112800624445
[6]LACOSTE E, MARTINEAU I, GAGNON G. (2003). Platelet concentrates: effects of calcium and thrombin on endothelial cell proliferation and growth factor release. J. Periodontol, 74(10): 1498-1507. https://doi.org/10.1902/jop.2003.74.10.1498
[7]XU J, GOU L, ZHANG P, et al. (2020). Platelet-rich plasma and regenerative dentistry. Aust. Dent. J., 65(2): 131-142. https://doi.org/10.1111/adj.12754
[8]TAYLOR G I, MINABE T. (1992). The angiosomes of the mammals and other vertebrates. Springer. Spec. Surg. S., 89(2): 181-215. https://doi.org/10.1097/00006534-199202000-00001
[9]Hua, W. M., Liang, Z. Q., Fang, Y, et al. (2009). Mechanism of curcumin against endothelial cell ischemia/reperfusion injury. Chinese Pharmacological Bulletin, 25(8), 1036-1041. https://portal.issn.org/resource/ISSN/1001-1978
[10]Xu, H. N., Xie, Z. Y., Qu, F. w., et al. (2024). Research progress on pretreatment methods to improve flap survival rate. Chinese Journal of Aesthetic and Plastic Surgery, 35(4), 247-250, 259. https://portal.issn.org/resource/ISSN/ 1673-7040
[11]LEE J H, YOU H J, LEE T Y, et al. (2022). Current Status of Experimental Animal Skin Flap Models: Ischemic Preconditioning and Molecular Factors. Int. J. Mol. Sci., 23(9): 5234. https://doi.org/10.3390/ijms23095234
[12]BOISSIERE F, GANDOLFI S, RIOT S, et al. (2021). Flap Venous Congestion and Salvage Techniques: A Systematic Literature Review. Plast Reconstr Surg Glob Open, 9(1): e3327. https://doi.org/10.1097/gox.0000000000003327
[13]HOU L, LI X, SU C, et al. (2022). Current Status and Prospects of Research on Ischemia-Reperfusion Injury and Ferroptosis. Front. Oncol, 12: 920707. https://doi.org/10.3389/fonc.2022.920707
[14]WANG B, GENG Q, HU J, et al. (2016). Platelet-rich plasma reduces skin flap inflammatory cells infiltration and improves survival rates through induction of angiogenesis: An experiment in rabbits. J Plast Surg Hand Surg, 50(4): 239-245. https://doi.org/10.3109/2000656x.2016.1159216
[15]OMORPHOS N P, GAO C, TAN S S, et al. (2021). Understanding angiogenesis and the role of angiogenic growth factors in the vascularisation of engineered tissues. Mol. Biol. Rep., 48(1): 941-950. https://doi.org/10.1007/s11033-020-06108-9
[16]Xuan, M. (2024). Experimental study on the involvement of autophagy in the healing of aging skin wounds [Doctoral dissertation, Southern Medical University]. Retrieved December 28, 2024, from CNKI Thesis Database. https://degree.nstl.gov.cn/thesis/1014335373
[17]AKBARZADEH S, MCKENZIE M B, RAHMAN M M, et al. (2021). Allogeneic Platelet-Rich Plasma: Is It Safe and Effective for Wound Repair? Eur Surg Res, 62(1): 1-9. https://doi.org/10.1159/000514223
[18]HENEWEER C, ZIRK M, SAFI A, et al. (2021). An Innovative Approach for Preoperative Perforator Flap Planning Using Contrast-enhanced B-flow Imaging. Plast Reconstr Surg Glob Open, 9(5): e3547. https://doi.org/10.1097/gox.0000000000003547
 Figures
Figures References
References Peer
Peer Information
InformationFigure 1 Flap design and fabrication schematic diagram.

Figure 2 Necrotic Area Analysis Diagram (X̅±s,%).

Figure 3 The schematic diagram of immunohistochemical staining results.

[1]KOUNIAVSKI E, EGOZI D, WOLF Y. (2022). Techniques and Innovations in Flap Engineering: A Review. Plast Reconstr Surg Glob Open, 10(9): e4523. https://doi.org/10.1097/gox.0000000000004523
[2]MYERS M B, CHERRY G. (1968). Causes of necrosis in pedicle flaps. Springer. Spec. Surg. S., 42(1): 43-50. https://pubmed.ncbi.nlm.nih.gov/4875691
[3]COLLINS T, ALEXANDER D, BARKATALI B. (2021). Platelet-rich plasma: a narrative review. EFORT Open Rev, 6(4): 225-235. https://doi.org/10.1302/2058-5241.6.200017
[4]XIE K, HUANG M, ZHENG Y, et al. (2022). Effect of Antilogous Platelet-Rich Plasma on the Revascularization of Rabbit Prefabricated Flap. Med Sci Monit, 28: e937718. https://doi.org/10.12659/MSM.937718
[5]YUAN T, GUO S C, HAN P, et al. (2012). Applications of leukocyte- and platelet-rich plasma (L-PRP) in trauma surgery. Current Pharm. Biotechnol,13(7):1173-1184. https://doi.org/10.2174/138920112800624445
[6]LACOSTE E, MARTINEAU I, GAGNON G. (2003). Platelet concentrates: effects of calcium and thrombin on endothelial cell proliferation and growth factor release. J. Periodontol, 74(10): 1498-1507. https://doi.org/10.1902/jop.2003.74.10.1498
[7]XU J, GOU L, ZHANG P, et al. (2020). Platelet-rich plasma and regenerative dentistry. Aust. Dent. J., 65(2): 131-142. https://doi.org/10.1111/adj.12754
[8]TAYLOR G I, MINABE T. (1992). The angiosomes of the mammals and other vertebrates. Springer. Spec. Surg. S., 89(2): 181-215. https://doi.org/10.1097/00006534-199202000-00001
[9]Hua, W. M., Liang, Z. Q., Fang, Y, et al. (2009). Mechanism of curcumin against endothelial cell ischemia/reperfusion injury. Chinese Pharmacological Bulletin, 25(8), 1036-1041. https://portal.issn.org/resource/ISSN/1001-1978
[10]Xu, H. N., Xie, Z. Y., Qu, F. w., et al. (2024). Research progress on pretreatment methods to improve flap survival rate. Chinese Journal of Aesthetic and Plastic Surgery, 35(4), 247-250, 259. https://portal.issn.org/resource/ISSN/ 1673-7040
[11]LEE J H, YOU H J, LEE T Y, et al. (2022). Current Status of Experimental Animal Skin Flap Models: Ischemic Preconditioning and Molecular Factors. Int. J. Mol. Sci., 23(9): 5234. https://doi.org/10.3390/ijms23095234
[12]BOISSIERE F, GANDOLFI S, RIOT S, et al. (2021). Flap Venous Congestion and Salvage Techniques: A Systematic Literature Review. Plast Reconstr Surg Glob Open, 9(1): e3327. https://doi.org/10.1097/gox.0000000000003327
[13]HOU L, LI X, SU C, et al. (2022). Current Status and Prospects of Research on Ischemia-Reperfusion Injury and Ferroptosis. Front. Oncol, 12: 920707. https://doi.org/10.3389/fonc.2022.920707
[14]WANG B, GENG Q, HU J, et al. (2016). Platelet-rich plasma reduces skin flap inflammatory cells infiltration and improves survival rates through induction of angiogenesis: An experiment in rabbits. J Plast Surg Hand Surg, 50(4): 239-245. https://doi.org/10.3109/2000656x.2016.1159216
[15]OMORPHOS N P, GAO C, TAN S S, et al. (2021). Understanding angiogenesis and the role of angiogenic growth factors in the vascularisation of engineered tissues. Mol. Biol. Rep., 48(1): 941-950. https://doi.org/10.1007/s11033-020-06108-9
[16]Xuan, M. (2024). Experimental study on the involvement of autophagy in the healing of aging skin wounds [Doctoral dissertation, Southern Medical University]. Retrieved December 28, 2024, from CNKI Thesis Database. https://degree.nstl.gov.cn/thesis/1014335373
[17]AKBARZADEH S, MCKENZIE M B, RAHMAN M M, et al. (2021). Allogeneic Platelet-Rich Plasma: Is It Safe and Effective for Wound Repair? Eur Surg Res, 62(1): 1-9. https://doi.org/10.1159/000514223
[18]HENEWEER C, ZIRK M, SAFI A, et al. (2021). An Innovative Approach for Preoperative Perforator Flap Planning Using Contrast-enhanced B-flow Imaging. Plast Reconstr Surg Glob Open, 9(5): e3547. https://doi.org/10.1097/gox.0000000000003547
Peer-review Terminology
Identity transparency: Single anonymized
Reviewer interacts with: Editor
Details
This is an open access article under the terms of the Creative Commons Attribution License(http://creativecommons.org/licenses/by/4.0/), which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Publication History
Received 2025-03-05
Accepted 2025-04-27
Published 2025-05-14


