Abstract

Pancreatic cancer demonstrates the greatest prevalence of thrombosis linked to
malignancy in solid tumors, accompanied by a markedly heightened risk of venous
thromboembolism (VTE). Early occurrence of VTE in cancer serves as a crucial predictor
of adverse prognosis, jeopardizing patient survival and quality of life. The
hypercoagulable condition triggered by the tumor microenvironment, integrated with host
genetic predisposition and treatment-associated hazards, collaboratively escalates VTE
susceptibility. This review methodically outlines the pathophysiology and elevated risk
elements for lower extremity deep vein thrombosis (DVT) in pancreatic cancer, while
appraising the efficacy of present preventive approaches, seeking to establish a
theoretical groundwork for refining clinical management.
Keywords:Pancreatic cancer; lower extremity deep vein thrombosis; risk factors; anticoagulant therapy; research progress
Introduction
Pancreatic cancer has emerged as the fourth leading cause of cancer-related mortality worldwide as of 2021, and its death rate is projected to rise to the second position by 2030 [1] . Despite the availability of various therapeutic strategies, the five-year survival rate for patients with pancreatic cancer remains dismally low, at only 10%. Notably, pancreatic cancer has the highest incidence of cancer-associated thrombosis (CAT) among all solid tumors, with the one-year cumulative incidence of VTE reaching as high as 7.4% following diagnosis [2,3] . Robust evidence from clinical studies has established that the occurrence of VTE in the early stages of malignancy is a critical prognostic indicator for poor outcomes in cancer patients [4] . Clinically, VTE primarily manifests as life-threatening pulmonary embolism (PE) and deep vein thrombosis (DVT), but may also include less common forms such as superficial vein thrombosis and catheter-related thrombosis. Beyond its direct threat to survival, VTE significantly impairs the quality of life in cancer patients [5] .
The progression of malignancy is a central driver of VTE risk, with a strong temporal association observed between the onset of metastatic disease and increased VTE incidence [6] . In pancreatic cancer, unique mechanisms of coagulation cascade activation not only accelerate the development of a prothrombotic state but also establish a bidirectional "thrombosis-tumor" regulatory network: a pathological coagulation microenvironment promotes tumor cell proliferation, while increasing tumor burden further elevates the risk of thrombosis [7] . This review aims to systematically analyze the pathophysiological mechanisms and high-risk factors underlying lower extremity deep vein thrombosis in patients with pancreatic cancer. Furthermore, we critically evaluate the clinical effectiveness of current preventive strategies based on evidence-based medicine, with the goal of providing a theoretical foundation for optimizing clinical management pathways.
Pathophysiological Mechanisms of Lower Extremity Thrombosis in Pancreatic Cancer
The dynamic remodeling of the tumor microenvironment (TME) in pancreatic cancer encompasses multiple processes, including extracellular matrix reorganization, immune cell recruitment, and aberrant angiogenesis. These changes, in concert with malignant tumor cells, activate Virchow's triad— comprising hypercoagulability, endothelial injury, and abnormal blood flow— through multidimensional interactions such as cytokine secretion, cell-cell contact, and mechanotransduction [8] . This activation significantly increases the clinical incidence of CAT and VTE.
In pancreatic cancer, a tightly regulated bidirectional network exists between CAT and the TME. CAT not only promotes tumor proliferation and invasion but is also potentiated by the TME, which enhances thrombotic propensity through alterations in the local microenvironment, such as hypoxia and acidosis. Together, these processes drive the malignant transformation toward a more aggressive phenotype, including epithelial-mesenchymal transition (EMT) and metastatic dissemination.
The TME and tumor cells collaboratively orchestrate the regulation of key molecules within the coagulation cascade, such as tissue factor, coagulation factor X, and thrombin. This regulation is mediated through multiple signaling pathways, including inflammatory pathways (e.g., NF-κB) and hypoxia-inducible signaling (e.g., HIF-1α), which contribute to the cascade of events underlying VTE pathogenesis—from initial thrombus formation to embolic progression [9] .
A prothrombotic state, characteristic of tumor tissue, plays a central role in this process. Its molecular basis involves the coordinated regulation of various inflammatory mediators (such as tissue factor, interleukin-6, and tumor necrosis factor-α) and procoagulant networks (including fibrinogen, von Willebrand factor, and platelet-activating factor). These factors form a positive feedback loop that further amplifies the risk of thrombosis.
Risk Factors for Lower Extremity Deep Vein Thrombosis in Pancreatic Cancer
Patient Characteristics
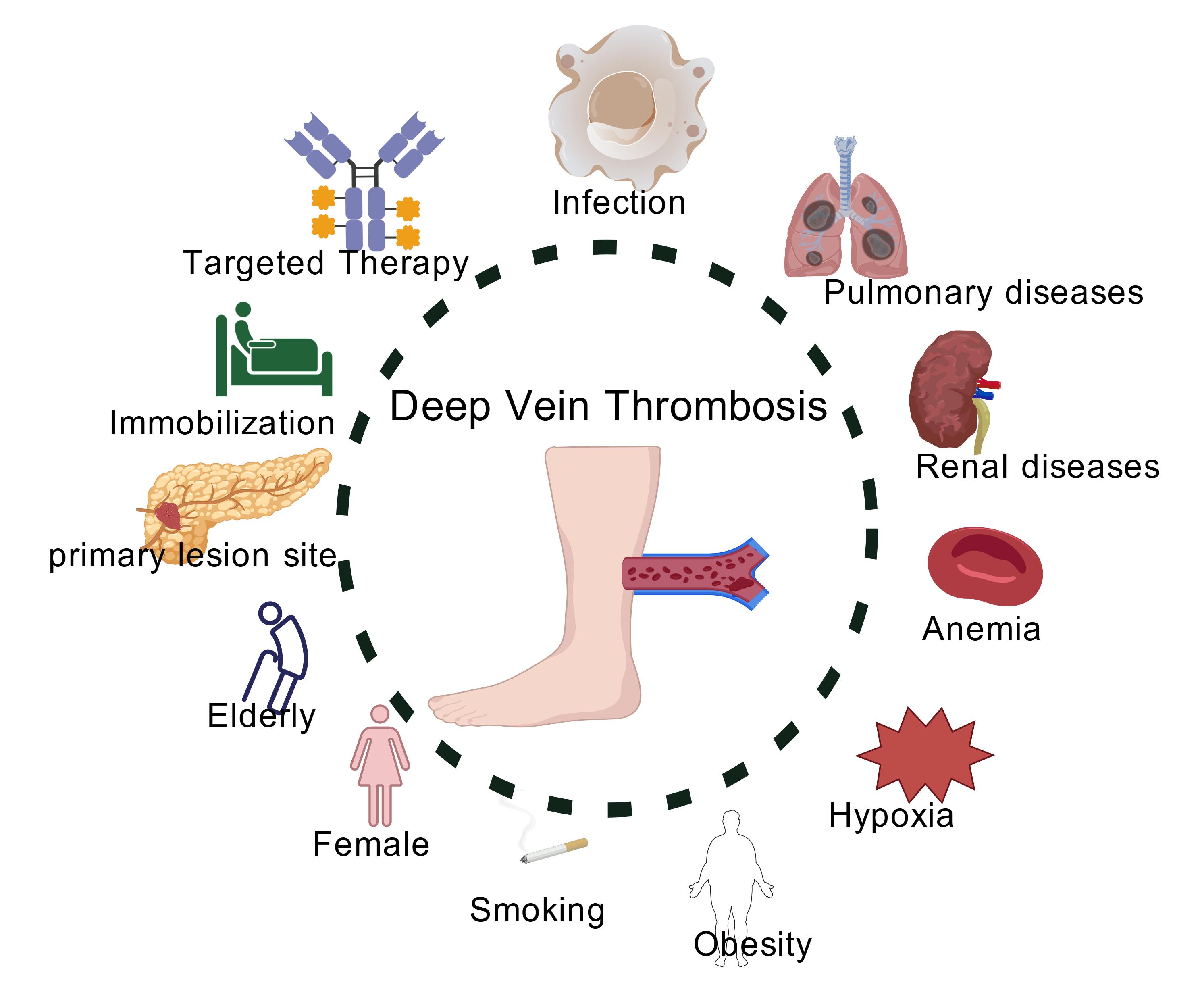
Patients with pancreatic cancer exhibit multidimensional and heterogeneous risk profiles for VTE [10] . The primary clinical risk factors include age over 65 years and reduced mobility, with immobilization (e.g., hospitalization with bed rest ≥72 hours) significantly increasing both the incidence and mortality of VTE in cancer patients [11,12] . Smoking is an independent risk factor for pancreatic cancer and further elevates the risk of VTE in patients with malignancies. Female patients with pancreatic cancer tend to have poorer VTE-related outcomes [13] . The presence of comorbidities such as obesity, pulmonary or renal disease, infection, or anemia synergistically increases the risk of VTE in patients with malignancies [14] . Both chemotherapy and hormone therapy are associated with a prothrombotic effect. Notably, the anatomical location of the primary tumor in the pancreas is correlated with thrombosis risk: tumors located in the body and tail of the pancreas are associated with a significantly higher risk of thrombosis compared to those in the pancreatic head.
Characteristic pathological changes in pancreatic cancer include extensive hypoxic regions (median tissue oxygen partial pressure 0–5.3 mmHg, markedly lower than the 24.3–92.3 mmHg observed in normal pancreatic tissue) and a dense fibrotic stroma. The latter acts as a physical barrier, exacerbating hypoxia and driving tumor progression. Hypoxia itself is recognized as an independent adverse prognostic factor in pancreatic cancer (Figure 1) [15] .
Figure 1. Amplifying VTE Risk Among PDAC Patients (created using BioRender.com).
Antitumor Therapy
Antitumor therapies for malignancies are generally associated with an increased risk of thrombosis, and standard treatment regimens for resectable pancreatic cancer—such as FOLFIRINOX or gemcitabine-based chemoradiotherapy— are inherently prothrombotic. Evidence-based studies have confirmed that chemotherapeutic agents including gemcitabine, 5-fluorouracil, and oxaliplatin have clear prothrombotic effects [16] . Chemotherapy-induced neutropenia often necessitates the use of hematopoietic growth factors for supportive care. Prospective cohort studies have demonstrated that the use of granulocyte colony-stimulating factor (G-CSF) significantly increases the risk of VTE in patients with pancreatic cancer. Additionally, the routine placement of central venous catheters during chemotherapy further elevates the risk of catheter-related thrombosis.
Major surgical procedures, such as pancreaticoduodenectomy, are associated with substantial tissue trauma and prolonged postoperative immobilization, leading to significant alterations in venous hemodynamics and an increased risk of blood stasis. In patients with pancreatic cancer complicated by chronic pancreatitis or pancreatic duct obstruction, the abnormal release of pancreatic enzymes may degrade anticoagulant proteins (e.g., antithrombin III) through proteolytic activity, thereby further exacerbating the hypercoagulable state.
Leukocytosis
Leukocytosis is significantly associated with an increased risk of VTE in patients with malignancies. A pre-chemotherapy white blood cell (WBC) count >11×10⁹/L has been established as an independent predictive factor in the Khorana risk score system. Neutrophils, the predominant component of peripheral WBCs, play a central role in the innate immune defense system. They contribute to thrombogenesis through the release of neutrophil extracellular traps (NETs) and, in conjunction with monocytes, the expression of the procoagulant tissue factor (TF) [17] , forming a dual pathway mechanism for leukocyte-mediated thrombosis. In pancreatic cancer patients, leukocytosis is associated with an increased risk of recurrent VTE, with aberrant NETs formation providing the pathophysiological basis for this association. In vitro studies have demonstrated that pancreatic cancer cell lines can induce NETs formation [18] . Furthermore, chemotherapy and targeted therapies can promote NETs release by damaging the endothelial cell surface [19] . Oto et al. identified seven microRNAs as potential predictive biomarkers for VTE in pancreatic cancer patients, and their study was the first to demonstrate the predictive potential of plasma microRNAs and neutrophil activation markers for VTE in this population [20] .
Platelet Hyperactivity
Platelet hyperactivity is considered a key mechanism underlying the hypercoagulable state observed in pancreatic cancer patients. Although platelets are primarily involved in arterial thrombosis, their role in venous thrombosis among cancer patients is increasingly recognized. Tumor-derived interleukin-6 (IL-6) stimulates hepatic thrombopoietin (TPO) synthesis, promoting thrombopoiesis and exacerbating thrombosis [21] . Studies have shown that the rate of thrombin generation in whole blood from pancreatic cancer patients is significantly higher than in plasma samples, with an overall increase in endogenous thrombin potential. These patients exhibit abnormally hyperreactive platelets, characterized by shortened degranulation times, all of which are associated with an increased risk of thromboembolic events [22] . Elevated levels of platelet activation markers are significantly correlated with increased VTE risk and reduced survival in pancreatic cancer patients. Platelet factor 4 (PF4) has been identified as a potential prognostic biomarker in pancreatic cancer, with elevated PF4 levels indicating poor prognosis and a significantly increased probability of VTE [23] . In addition, P-selectin, a member of the selectin family, is expressed on the surface of platelets and endothelial cells during coagulation cascade activation and serves as a key plasma marker of coagulation activation by promoting platelet aggregation [24] . Clinical studies have demonstrated that elevated P-selectin levels are significantly associated with increased VTE risk and mortality in pancreatic cancer patients [25] . As P-selectin is expressed by both endothelial cells and platelets, its dual expression can enhance VTE risk by recruiting leukocytes. Antiplatelet agents such as aspirin and clopidogrel may contribute to VTE prevention in certain cancer populations.
Coagulation Factor Dysregulation
Patients with pancreatic cancer exhibit significant elevations in plasma levels of kallikrein complexes and coagulation factors IX, XIIa, and Xa, as well as their respective natural inhibitor complexes, indicating aberrant activation of the intrinsic coagulation pathway [26] . Typically, these patients present with increased levels of fibrinogen, factor VIII (FVIII), and D-dimer, accompanied by decreased activity of protein C and antithrombin III. Tumor-derived signaling molecules such as interleukin-1 (IL-1), vascular endothelial growth factor (VEGF), and tumor necrosis factor-alpha (TNF-α) collectively drive a bidirectional network of inflammation and coagulation [27] .
Tissue factor, a glycosylated transmembrane receptor protein, specifically binds and activates plasma coagulation factor VII (FVII). The TF/FVIIa complex serves as the critical initiator of the extrinsic coagulation cascade [28] . Numerous studies have demonstrated aberrant overexpression of TF in various malignancies, with TF abundance in pancreatic tumor tissue being closely associated with VTE incidence. Tumor-derived TF can enter the circulation via secretion of TF-positive extracellular vesicles (TF+EVs) [29] . Clinical data indicate that plasma TF+EVs concentrations are significantly correlated with VTE occurrence in pancreatic cancer patients [30] , suggesting a pivotal role for circulating TF+EVs in pathological thrombogenesis.
In pancreatic cancer, procoagulant proteins released in microparticle or non-microparticle forms not only accelerate thrombus formation but also amplify inflammatory responses. These microparticles upregulate the expression of intercellular adhesion molecules, enabling direct participation of tumor cells in the coagulation cascade and triggering both local and systemic coagulation activation [31] . High-molecular-weight mucins secreted by pancreatic cancer cells enter the circulation, with their concentrations positively correlated with tumor progression. Animal model studies have confirmed their biological effect in inducing microthrombus formation [32] . The natural anticoagulant protein S (PS) is markedly downregulated during pancreatic cancer progression [33] . Approximately 60% of circulating PS is inactivated by binding to C4b-binding protein (C4BP), leaving only 40% in the free, functionally active form; thus, measurement of free PS levels is a key indicator for thrombosis risk assessment [34] . Studies in obese animal models have shown that hepatic PS levels are significantly reduced under hypoxic conditions, providing a theoretical basis for targeting PS to simultaneously improve the tumor microenvironment and reduce thrombotic risk [35] .
Plasminogen activator inhibitor-1 (PAI-1), a member of the serine protease inhibitor family, inhibits both tissue-type plasminogen activator (tPA) and urokinase-type plasminogen activator (uPA), thereby significantly reducing plasmin generation and impairing fibrinolytic function. Chronic inflammation within the tumor microenvironment upregulates PAI-1 expression in host endothelial cells, resulting in a fibrinolytic inhibitory state. This pathophysiological mechanism not only enhances thrombus stability but also promotes thrombogenesis by reducing fibrin degradation. PAI-1 is primarily synthesized by endothelial cells, and its plasma levels are markedly elevated in conditions of acute or chronic inflammatory endothelial injury, correlating positively with thrombotic risk [36] . Recent clinical research by Hisada et al. confirmed that elevated plasma PAI-1 activity in pancreatic cancer patients is significantly associated with increased VTE incidence [37] . These findings suggest that pharmacological inhibition of PAI-1 may represent a novel therapeutic strategy for preventing cancer-associated thrombosis.
Von Willebrand factor (VWF), a large multimeric glycoprotein, plays a central role in physiological hemostasis by mediating platelet adhesion to subendothelial collagen and facilitating platelet-platelet interactions under high shear stress conditions. VWF is synthesized by endothelial cells and megakaryocytes and stored as large multimers in Weibel-Palade bodies (WPBs) of endothelial cells and α-granules of platelets [38] . Within the tumor microenvironment, tumor cells can induce endothelial VWF release, and some tumor cells are capable of de novo VWF synthesis, thereby enhancing platelet adhesion and activation and ultimately promoting pathological thrombosis [39] . Clinical studies have demonstrated that elevated plasma VWF levels in cancer patients are significantly associated with advanced tumor stage, poor prognosis, and serve as an independent risk factor for VTE. A cohort study enrolling 194 cancer patients between January 2013 and September 2018 revealed that VWF levels in patients with gastric, esophageal, colorectal, and pancreatic cancers were significantly higher than those in healthy controls [40] .
Abnormal coagulation parameters in pancreatic cancer have important clinical implications. Elevated CA19-9 levels have been identified as a core risk factor for VTE in pancreatic cancer patients, and VTE events are closely associated with worsened overall prognosis. Moreover, the severity of VTE correlates positively with CA19-9 concentrations, with pancreatic cancer patients experiencing VTE exhibiting significantly higher CA19-9 levels than those without thrombosis [41] . Patients with elevated platelet counts have a higher incidence of thrombotic events compared to those with normal platelet counts, and those with abnormal PF4 levels prior to chemoradiotherapy are at even greater risk for VTE [42] (Table 1).
Table 1. Key Indicators and Their Relationship with Pancreatic Cancer and VTE
|
Indicator Name |
Abbreviation |
Relationship with Pancreatic Cancer and VTE |
|
Protein S |
PS |
Animal models indicate that reduced hepatic PS levels are associated with hypoxia, providing a theoretical basis for targeted interventions to improve the tumor microenvironment and reduce thrombosis risk. |
|
Plasminogen Activator Inhibitor-1 |
PAI-1 |
Elevated plasma PAI-1 activity in pancreatic cancer patients is significantly associated with increased VTE incidence; targeted inhibition may represent a novel therapeutic strategy for cancer-associated thrombosis prevention. |
|
von Willebrand Factor |
VWF |
VWF levels are significantly higher in pancreatic cancer patients compared to healthy controls; elevated VWF is an independent risk factor for VTE and is associated with advanced tumor stage and poor prognosis. |
|
Carbohydrate Antigen 19-9 |
CA19-9 |
Elevated CA19-9 is a key risk factor for VTE in pancreatic cancer patients; the severity of VTE is positively correlated with CA19-9 concentration, and levels are significantly higher in patients with VTE compared to those without. |
|
Platelet Count |
— |
Increased platelet count is associated with a higher incidence of thrombotic events compared to normal levels. |
|
Platelet Factor 4 |
PF4 |
Abnormal pre-chemoradiotherapy PF4 levels are associated with a significantly increased risk of VTE. |
VTE Prophylaxis in Patients with Pancreatic Cancer
For high-risk patients undergoing systemic anti-tumor therapy—such as those with advanced pancreatic cancer—the International Society on Thrombosis and Haemostasis (ISTH) guidelines recommend outpatient thromboprophylaxis with therapeutic-dose low molecular weight heparin (LMWH). However, traditional anticoagulation regimens are limited by the risk of bleeding complications, particularly when combined with cytotoxic chemotherapy agents [43] . In patients with locally advanced or metastatic pancreatic cancer, the CONKO-004 and FRAGEM randomized controlled trials evaluated the efficacy of different LMWH dosing strategies [44,45] . The CONKO-004 trial utilized a high prophylactic dose of enoxaparin (1 mg/kg qd), while the FRAGEM study employed a weight-adjusted dose of dalteparin (200 IU/kg qd). Both studies demonstrated that anticoagulation significantly reduced the incidence of VTE without a corresponding increase in major bleeding events (p > 0.05).
For patients undergoing curative pancreatic tumor resection, strict adherence to standardized perioperative LMWH prophylaxis is essential. Recent guidelines also recommend the use of novel oral factor Xa inhibitors (such as apixaban and rivaroxaban) for thromboprophylaxis in hospitalized cancer patients and outpatients receiving chemotherapy. Individualized anticoagulation strategies should be developed based on risk stratification tools such as the Khorana score [46] . The CASSINI and AVERT multicenter trials assessed the efficacy and safety of prophylactic-dose direct oral anticoagulants (DOACs) in outpatients receiving chemotherapy with a Khorana score ≥2 [47] . Subgroup analysis from the CASSINI trial showed that, among 273 ambulatory patients with pancreatic cancer at various stages, the incidence of venous thromboembolism was significantly lower in the rivaroxaban group (hazard ratio=0.35, 95% confidence interval 0.13–0.97), with no statistically significant difference in treatment-related bleeding events compared to placebo (p=0.18). A retrospective analysis of 130 patients in the AVERT trial confirmed that prophylactic apixaban significantly reduced VTE incidence in patients with gastrointestinal malignancies, with no significant difference in bleeding rates between groups (p=0.32). A meta-analysis focusing on ambulatory pancreatic cancer patients evaluated the clinical value of anticoagulants for primary VTE prevention during chemotherapy. This study integrated data from five international clinical trials, including FRAGEM and CONKO-004, comparing the efficacy of LMWH and DOACs. The pooled analysis demonstrated a significantly lower VTE incidence in the prophylactic anticoagulation group compared to controls (4% vs. 12%), confirming both the safety and efficacy of thromboprophylaxis during chemotherapy.
Chemotherapy-induced thrombocytopenia (CIT) is common in the clinical management of pancreatic cancer. Chemotherapeutic agents not only suppress bone marrow hematopoiesis, leading to reduced platelet counts, but also cause toxicities such as mucositis, significantly increasing the risk of bleeding. Multicenter clinical data indicate that the incidence of grade 3–4 thrombocytopenia is up to 9% with FOLFIRINOX for locally advanced or metastatic pancreatic cancer, 4–9% with gemcitabine monotherapy, and 4–13% with gemcitabine plus nab-paclitaxel [48,49] . Tumor-infiltrative biological features may further exacerbate bleeding tendencies, primarily through invasive lesions of the gastroduodenal wall and secondary portal hypertension [50] . According to EHA guidelines, patients with grade 1–2 thrombocytopenia (platelet count 50–100 × 10⁹/L) may continue standard-dose LMWH, apixaban, or rivaroxaban; for grade 3 (25–50 × 10⁹/L), DOACs should be discontinued, and LMWH may be used cautiously if there is no bleeding and platelet counts are stable; for grade 4, all anticoagulation should be stopped and only resumed when platelet counts recover to >50 × 10⁹/L. All patients receiving systemic anti-tumor therapy should be co-administered proton pump inhibitors (PPIs) to prevent gastrointestinal bleeding.
Due to the hypercoagulable state and complex surgical trauma, postoperative VTE risk is significantly increased in pancreatic cancer patients, particularly those undergoing vascular resection and reconstruction, where the incidence of portal-mesenteric vein thrombosis is notably high. The incidence of postpancreatectomy hemorrhage (PPH) in these patients ranges from 7.6% to 12.3% [51] , necessitating a careful risk-benefit assessment for thromboprophylaxis. A multicenter survey by Groen et al. involving 167 pancreatic surgeons revealed significant heterogeneity in thromboprophylaxis protocols across institutions (over 10 different regimens), with 39% of surgeons modifying anticoagulation strategies after vascular resection [52] . A systematic review of 27 studies on anticoagulation and 10 on non-anticoagulation regimens found marked diversity in institutional protocols and significant differences in vascular reconstruction techniques among intervention groups. Notably, the 30-day postoperative VTE incidence was higher in the anticoagulation group (5%) compared to the non-anticoagulation group (2%), while the incidence of PPH was similar in both groups (7%). The review concluded that current evidence is insufficient to establish a definitive clinical pathway for postoperative anticoagulation in pancreatic cancer [53] . Given the unique tumor biology and perioperative hypercoagulable state in patients undergoing pancreatic resection, the risk-benefit balance of extended thromboprophylaxis may differ substantially. Several systematic reviews focusing on postoperative anticoagulation strategies in pancreatic cancer have demonstrated that extending LMWH prophylaxis to 28–35 days postoperatively can reduce VTE incidence by approximately 58% without compromising safety. However, due to the lack of high-level evidence, current clinical guidelines have yet to provide clear recommendations for postoperative thromboprophylaxis in this patient population (Table 2).
Table 2. VTE Prevention Strategies for Different Patient Categories
|
Population Category |
VTE Prevention Strategy |
Evidence/Guideline Support |
|
High-risk patients receiving systemic anti-tumor therapy (advanced/metastatic) |
1. Therapeutic-dose LMWH: Enoxaparin 1 mg/kg qd, Dalteparin 200 IU/kg
qd; |
1. CONKO-004, FRAGEM studies (reduced VTE, no increase in major
bleeding); |
|
Patients undergoing curative pancreatic resection |
1. Perioperative: Standardized LMWH; |
1. Guidelines recommend perioperative standardized LMWH; |
|
Patients with chemotherapy-induced thrombocytopenia (CIT) |
1. Grade 1–2 (platelets 50–100×10⁹/L): Maintain standard-dose
LMWH/DOAC; |
EHA guideline recommendation |
Discussion
The pathophysiological process of pancreatic cancer is characterized by enhanced procoagulant activity, impaired anticoagulant function, and dysregulated fibrinolysis, collectively contributing to a prothrombotic state that underpins its core mechanism. Extensive research has established a strong association between pancreatic cancer and VTE, with VTE serving as an independent risk factor for poor prognosis in these patients. This relationship is primarily driven by the hypercoagulable state induced by the tumor microenvironment, which manifests as abnormal hematological parameters, excessive platelet activation, and disruption of the balance between coagulation and anticoagulation systems. These changes promote both local and systemic coagulation responses.
Host genetic susceptibility, in conjunction with acquired risk factors related to pancreatic cancer treatment, further amplifies the incidence of VTE. The Khorana risk score provides a quantitative framework for assessing thrombosis risk in patients undergoing pancreatic resection and is applicable to all patients, regardless of tumor resectability. Surgical candidates require heightened vigilance for both postoperative thrombosis and PPH.
Currently, research into the interplay between pancreatic cancer and VTE faces significant challenges in translating the complex bidirectional regulation between tumor biology and the coagulation system into clinical practice. There is an urgent need to integrate multi-omics data analysis and conduct prospective studies to develop a precise and individualized strategy for VTE prevention and management in pancreatic cancer patients.
Conclusion
Lower extremity deep vein thrombosis in pancreatic cancer—the malignancy with the highest incidence of thrombotic events—results not from a single factor but from systematic activation of Virchow's triad through interactions among the tumor microenvironment, therapeutic interventions, and patient-specific factors. This review systematically analyzes the underlying pathophysiological mechanisms, revealing that hypoxia, inflammation, and stromal hypertension within the tumor microenvironment drive a cascade regulation of tissue factor, inflammatory mediators, and coagulation factors, establishing a reciprocal "thrombosis–tumor" regulatory network. Patient characteristics such as advanced age, immobility, and obesity, combined with antitumor therapies including chemotherapy, radiotherapy, and surgery, further potentiate this hypercoagulable state.
We also critically evaluate current evidence-based preventive strategies. High-quality clinical evidence demonstrates that prophylactic anticoagulation significantly reduces the incidence of venous thromboembolism in patients receiving systemic therapy for advanced pancreatic cancer, without a significant increase in major bleeding. However, chemotherapy-induced thrombocytopenia and tumor-associated bleeding tendencies remain major limitations to anticoagulation use. In patients undergoing curative resection, the coexistence of postoperative hypercoagulability and elevated bleeding risk underscores the lack of standardized anticoagulation protocols; current evidence suggests potential benefits from extended low molecular weight heparin prophylaxis, though higher-level evidence is needed.
In summary, the prevention and management of lower extremity deep vein thrombosis in pancreatic cancer has entered an era of risk-stratified, personalized precision medicine. Future research should focus on developing novel anticoagulants with improved efficacy and safety profiles, validating predictive biomarkers and clinical risk models, and establishing standardized anticoagulation strategies for high-risk settings such as the perioperative period. Integrating pathophysiological insights, risk prediction, and individualized intervention holds promise for optimizing clinical management pathways—effectively reducing thrombotic events while maximizing patient safety and improving survival and quality of life.
Abbreviations
C4BP: C4b-Binding Protein; CAT: Cancer-Associated Thrombosis; CIT: Chemotherapy-Induced Thrombocytopenia; DVT: Deep Vein Thrombosis; EMT: Epithelial-Mesenchymal Transition; IL-1: Interleukin-1; ISTH: International Society on Thrombosis and Haemostasis; LMWH: Low Molecular Weight Heparin; PAI-1: Plasminogen Activator Inhibitor-1; PF4: Platelet Factor 4; PPH: Postpancreatectomy Hemorrhage; PS: Protein S; PPIs: Proton Pump Inhibitors; TF+EVs: TF-Positive Extracellular Vesicles; TPO: Thrombopoietin; tPA: Tissue-Type Plasminogen Activator; TME: Tumor Microenvironment; TNF-α: Tumor Necrosis Factor-α; uPA: Urokinase-Type Plasminogen Activator; VEGF: Vascular Endothelial Growth Factor; VTE: Venous Thromboembolism; VWF: Von Willebrand Factor; WPBs: Weibel-Palade Bodies.
Declarations
Author contributions
Junjie Zhao: Drafting of the manuscript, preparation, creation, and presentation of the published work, specifically writing the initial draft, including substantial translation. Huaping Wu: Supervision, oversight, and leadership responsibility for the planning and execution of the research activities. All authors have read and approved the final manuscript.
Acknowledgement
Not applicable.
Funding information
Not applicable.
Ethics approval and consent to participate
Not applicable.
Competing Interests
The authors declare that they have no existing or potential commercial or financial relationships that could create a conflict of interest at the time of conducting this study.
Data Availability
Not applicable.
References
 Figures
Figures References
References Peer
Peer Information
InformationFigure 1. Amplifying VTE Risk Among PDAC Patients (created using BioRender.com). Characteristic pathological changes in pancreatic cancer include extensive hypoxic regions and a dense fibrotic stroma, which acts as a physical barrier to exacerbate hypoxia and drive tumor progression; hypoxia itself is an independent adverse prognostic factor and amplifies the risk of venous thromboembolism.

Peer-review Terminology
Identity transparency: Single anonymized
Reviewer interacts with: Editor
Details
This is an open access article under the terms of the Creative Commons Attribution License(http://creativecommons.org/licenses/by/4.0/), which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Publication History
Received 2025-09-23
Accepted 2026-01-29
Published 2026-04-06


